Israel-based medical device company CartiHeal’s Agili-C™ implant is indicated for the treatment of knee-joint surface lesions (JSLs). It is the only approved implant that promotes natural articular cartilage and subchondral bone repair.
The US Food and Drug Administration granted premarket approval (PMA) for the implant in March 2022. The FDA previously awarded breakthrough device designation for the implant in 2020.
The implant received a CE mark from the European Commission in 2011.
Details of joint surface lesions (JSLs)
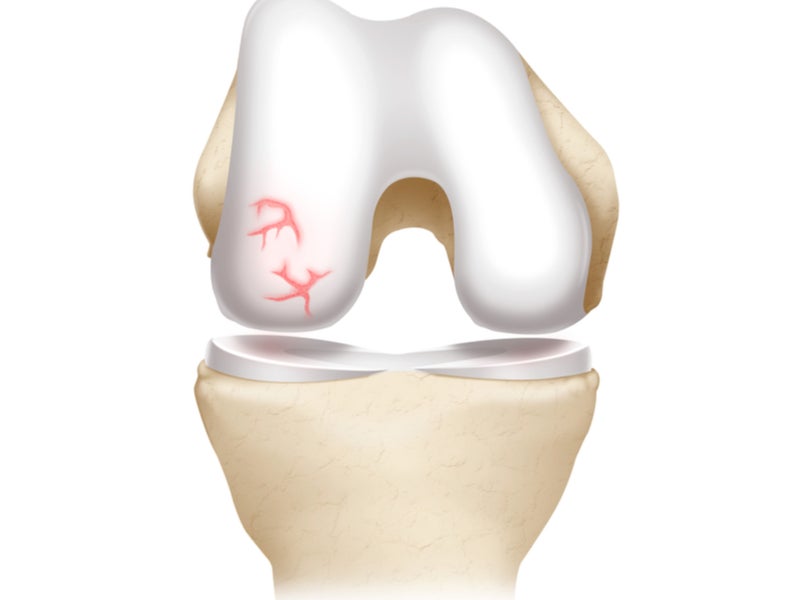
Articular cartilage and subchondral bone are the two primary tissues that constitute the knee joint. These tissues work together to provide a load-bearing mechanism that allows the joint to move freely and painlessly.
The subchondral knee joint is protected by articular cartilage, which absorbs stress, distributes load, and allows for smooth and stable mobility.
JSLs of the articular cartilage and subchondral bone are a common orthopaedic condition that lead to partial-thickness cartilage defects. JSLs can cause considerable pain and dysfunction of the knee joint and comprise 20% of all arthroscopic procedures.
The condition can lead to further cartilage loss and even osteoarthritis if left untreated, mainly due to the poor self-healing ability of the cartilage.
Features of the Agili-C implant
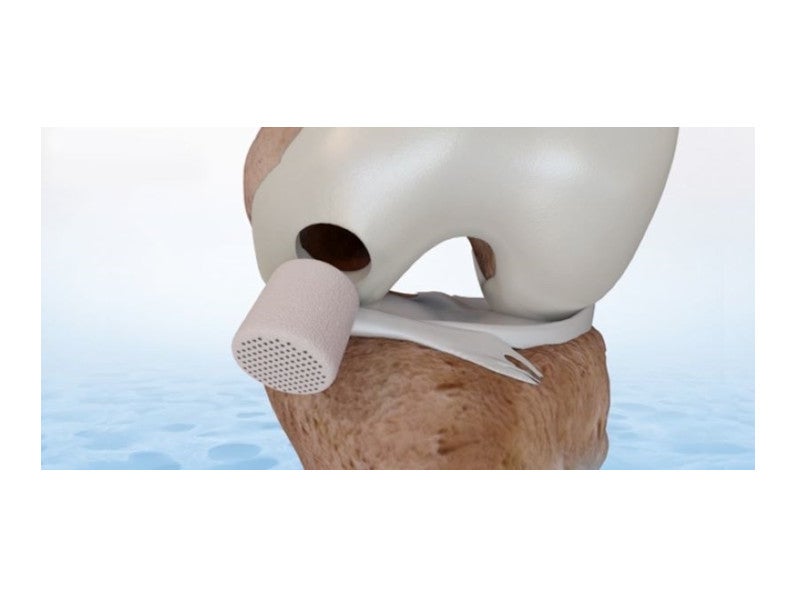
The Agili-C implant is a bi-phasic, porous and resorbable scaffold made from natural calcium carbonate, aragonite, which is derived from coral exoskeleton. It promotes the regeneration of articular cartilage and subchondral bone through a natural process without the use of cells or growth factors.
The implant stimulates the migration of chondrocytes cells, which are responsible for cartilage formation, into the porous structure of the scaffold with the aim to reproduce the original structure and function of the articular joint.
The Agili-C implant is recommended for the treatment of knee JSLs of International Cartilage Repair Society (ICRS) grade III or above, with a total targeted area of 1-7cm2 and without severe osteoarthritis (Kellgren-Lawrence grade 0-3).
Implantation procedure for Agili-C
The implantation procedure starts by identifying the size of the defect, after which a hole is drilled in the afflicted area. The implant is then pressed into place in the prepared hole. Blood quickly infiltrates the connected scaffold pores following implantation.
Two processes occur simultaneously during implant degradation. The first of these is bone remodelling by osteoclasts and osteoblasts, while the second is new cartilage regeneration by cell adhesion and development of the extracellular matrix of the hyaline cartilage.
Clinical studies on the Agili-C implant
The PMA for the implant was granted based on the results of a two-year multicentre, randomised, two-arm, open-label IDE clinical trial, which enrolled 251 people at 26 locations across the US, Europe and Israel. The patients enrolled were aged between 21 and 75 years and had up to three JSLs.
The clinical trial compared Agili-C to the Surgical Standard of Care (SSOC), including treatment of lesions using arthroscopic standard procedures such as microfracture and debridement.
The study’s primary endpoint was the change in the average Knee Injury and Osteoarthritis Outcome Score (KOOS) from baseline to 24 months. This comprised five subscales, namely pain, other symptoms, quality of life (QOL), activities of daily living (ADL), and sports. The KOOS Overall Score ranges from zero to 100, with higher values indicating better results.
The study found that the mean of the posterior distribution for changes from baseline to 24 months in the KOOS overall score for patients who were randomised to Agili‑C was 42.65, compared with 21.39 for patients randomised to SSOC.
The results of the study revealed that Agili-C was superior to the current SSOC after 24 months, with the Bayesian posterior probability of superiority estimated to be 1.00 in the Agili-C arm, exceeding the predetermined superiority threshold of 0.98.
In addition, the study revealed that 88.5% of patients treated with Agili‑C had at least 75% of their cartilage defect filled with new tissue, compared with 30.9% of patients in the SSOC group.
The study’s secondary endpoints were pain, function in daily living and QOL. These confirmed the Agili-C implant’s superiority over SSOC.
Marketing commentary on CartiHeal
CartiHeal is a medical device company based in Israel and the US. It produces unique implants for the treatment of traumatic and osteoarthritic joint cartilage and osteochondral abnormalities.