Koning Vera Breast CT (KBCT) is a 3D breast imaging device developed by Koning Health, a medical device company based in the US.
It produces real 3D images of the breast, enabling pain-free and early detection of cancers without breast compression.
The patented device provides isotropically accurate images of the breast, as well as diagnostic and biopsy-guided examinations, both with and without the utilisation of contrast.
It is said to be more accurate than mammography, tomosynthesis, and other traditional methods.
The device is currently available in more than 20 locations worldwide.
Koning Vera Breast CT launch and installations
Brookwood Baptist Medical Center in Alabama, US, installed the first Koning Vera Breast CT device, which became available to the public in January 2023.
The device was showcased at the Radiological Society of North America’s annual meeting in December 2023.
Koning collaborated with Gentle Scan Health, a venture company, in December 2023 to install 20 Koning Vera Breast CT devices across the US in 2024, starting from the Metropolitan New York City area, Atlanta, and Southern California.
The company also partnered with Chesa Industries to install ten KBCTs in clinics across Southeast Asia in October 2021.
Swissray, a medical imaging devices developer based in the US, is one of Koning’s distribution partners for the device.
Regulatory approvals for Koning Vera Breast CT
The initial cone beam computed tomography (CBCT) version of the device received a CE mark in Europe in February 2012.
Koning Breast CT (model KBCT 1000) received premarket approval market clearance from the US Food and Drug Administration (FDA) in February 2015.
The enhanced commercial version of the device received FDA approval in 2017. The American Medical Association authorised the use of the dedicated breast CT CPT code for diagnostic purposes in January 2020.
The commercial version of the KBCT device received approval in China in 2018. The device has also been approved in Canada, Singapore, Australia, Thailand, and Qatar.
Koning Vera Breast CT details and features
Koning Vera Breast CT is a low-dose, 3D scanner that can capture images of the entire breast in a single scan. It is claimed to be the only device with a true 360-degree scanning angle.
It is a small device that does not require painful compression of the breasts. The scan takes just seven seconds and exposes the patient to low levels of radiation, similar to a diagnostic mammogram.
It has a large field of view, which can cover up to 34cm longitudinally, including the chest wall.
The device can detect stage zero and stage one cancers and identify small lesions of 2mm in size and 200-micron calcifications. It can identify potential abnormalities with high precision for accurate diagnosis.
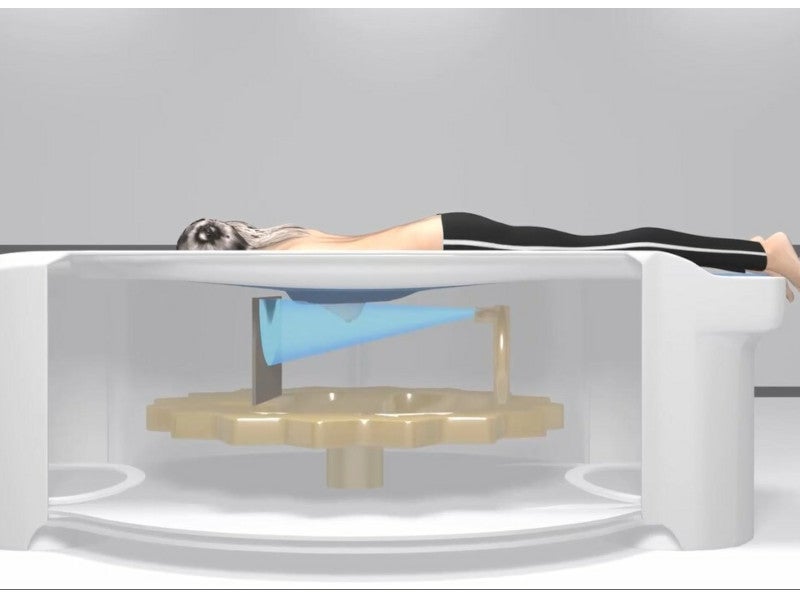
During the scan, the patient is made to lie down on a table facing downwards. The imaging device rotates about the breast once for seven seconds while the other breast lies suspended in the open.
The detector surrounds the woman’s breast to identify cancerous tissue from every angle for early disease detection, diagnosis, intervention, and treatment.
The resulting 3D images provide a clear and detailed view of the breast tissues with high spatial resolution.
Moreover, the device features a 3D biopsy capability with an average biopsy time of approximately 15 minutes.
Koning Vera Breast CT benefits
The Koning Vera Breast CT device is an affordable solution for breast imaging with high throughput. Its improved spatial resolution improves the way clinicians visualise and assess breast tissue.
It helps in the early detection of small tumours and offers a low implant rupture risk, making it ideal and safe for patients with breast implants.
It provides an efficient breast density distribution and ensures clear visualisation, further helping in detecting calcification clusters called ductal carcinoma in situ.