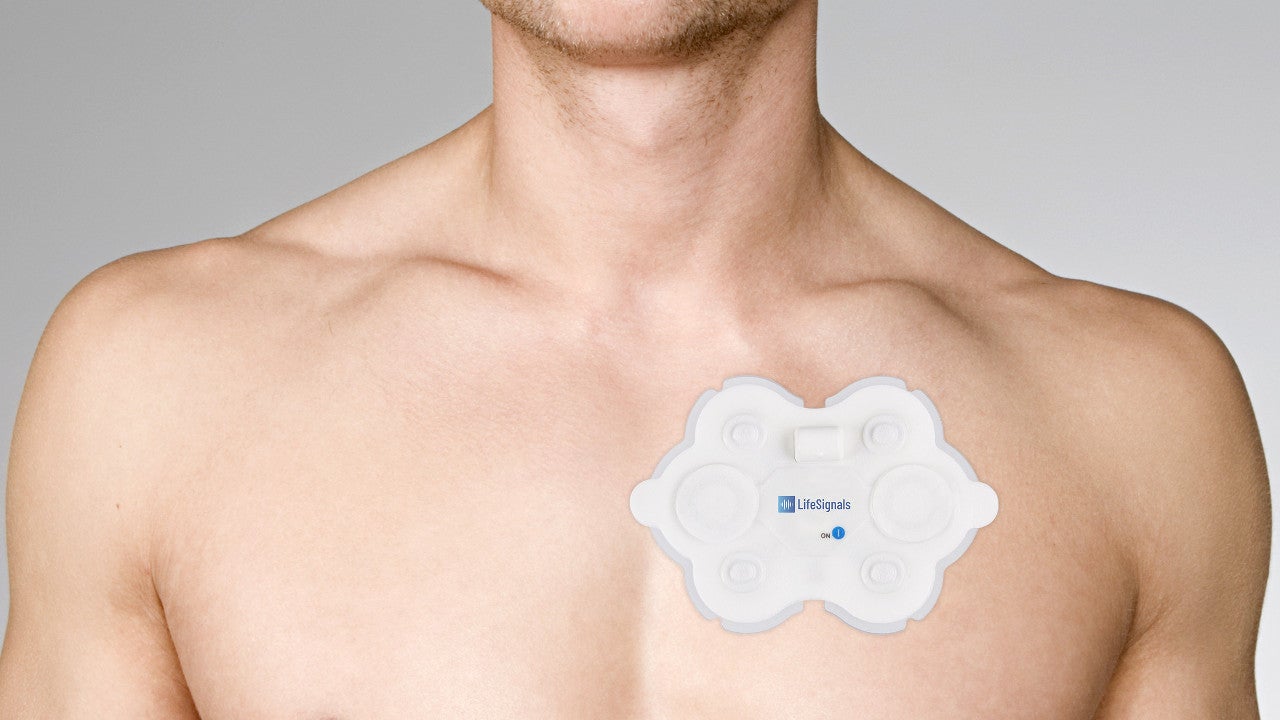
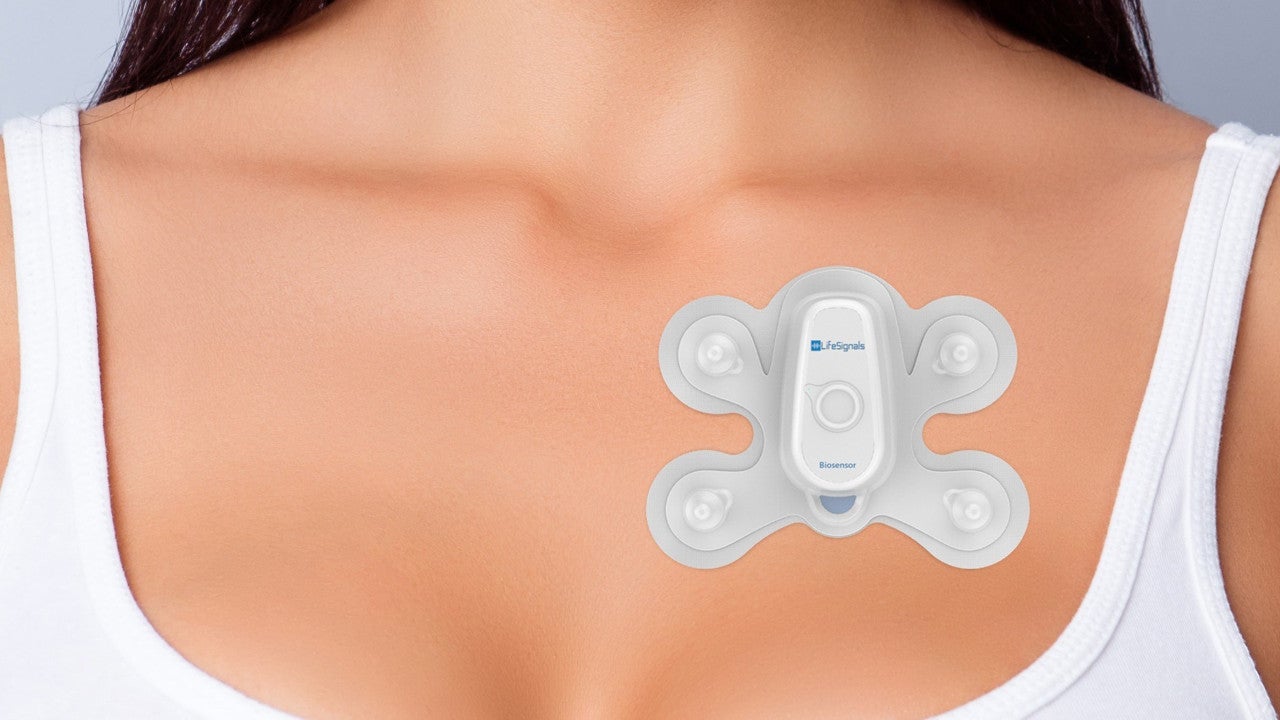
LP1250 is a wireless remote monitoring system developed by LifeSignals Group to allow healthcare professionals to continuously collect electrocardiography (ECG) and monitor the heart rate of patients in various care settings including home, hospital and ambulance.
LifeSignals Group was formed by a merger between LifeSignals Inc’s LifeSignals portfolio and Renew Health Limited’s connected health portfolio in January 2020.
A wireless remote monitoring system will enable remote patient monitoring services for rapid treatment decision-making and render data-driven, tailored treatment to the patients.
The system was approved by the Health Sciences Authority (HSA) of Singapore in April 2020, while the European CE Mark certification was granted in May 2020.
The US Food and Health Administration (FDA) cleared the system for human use in July 2020. The system will be available as a white-labelled device worldwide via partnerships among OEMs, telehealth software providers and hospitals.
Details of LP1250 wireless remote monitoring system
LP1250 ECG remote monitoring system is the first single-use, two-channel ECG and heart rate monitor in the world. It is a wearable, interoperable, ready-to-integrate biosensor, which diagnoses multiple parameters and allows fast onboarding.
LP1250 biosensor captures 72-hour ECG and heart rate data for improved diagnosis of patients, equipped with the proprietary single-chip LC1100 Life Signal™ Processor platform, which facilitates secured diagnosis, storage and transmission of data in any care setting.
The single-use device decreases the risk of infection transmission and operational expenditure. The lightweight and splash-proof tool increases comfort and compliance in patients. It transmits the data wirelessly to the remote secure server for storage and analysis. The physicians can access the patient data remotely through third-party software for the monitoring of common cardiac arrhythmias, irrespective of the patient’s location.
LC1100 Life Signal Processor features
LC1100 is silicon, single-chip solution used in disposable, rechargeable, biosensor patches of clinical grade.
A fully configurable chip integrates multiple sensor interfaces, multiple radios, application processor and power management for enabling signal acquisition, processing, wireless transmission and storage capabilities. It acquires signals with high precision from various biosensors including respiration, temperature, pulse-oximetry and ECG.
The multi-mode radio is based on the 2.4GHz Wi-Fi standard, 2.36-2.4GHz Medical Band (MBAN) and a 3-10GHz Ultra-Wideband (UWB) based transmitter.
The chip is integrated with digital interfaces to sense motion, audio and external data storage. It consumes low power and offers high link reliability.
LC1100 is highly immune to electromagnetic interference by intelligently switching between multi-band radios. It uses 2V to 3.6V batteries for complete on-chip power management, which provides power for up to five days. Firmware is embedded on a companion flash chip for Wi-Fi and radio management.
It utilises ARM Cortex-M0 application processor to personalise analytics and system operation for original equipment manufacturers (OEMs).
Details of Life Signal patch
Life Signal™ patch is a fully disposable low-power, multi-parameter biosensor worn on the chest for physiological data collection in various settings.
It is a lightweight patch, weighing under two grams and measuring 110mm x 80mm x 6mm. The patch offers wireless connection range of up to 25m.
LifeSignals’ wireless biosensor patches for COVID-19
LifeSignals is accelerating the introduction of single-use biosensor patch 1AX for early identification and monitoring of symptoms in COVID-19 patients. The company is currently producing the wireless medical biosensor patch in high volume.
The biosensor patch 1AX, based on a proven cardiovascular monitoring platform, can be fixed on the chest to record real-time temperature, respiration, ECG, heart rate and movement-related data.
Data is then transmitted from the patch wirelessly to a mobile application on the user’s phone in real-time and can be used for effective remote screening of the patients in quarantine.