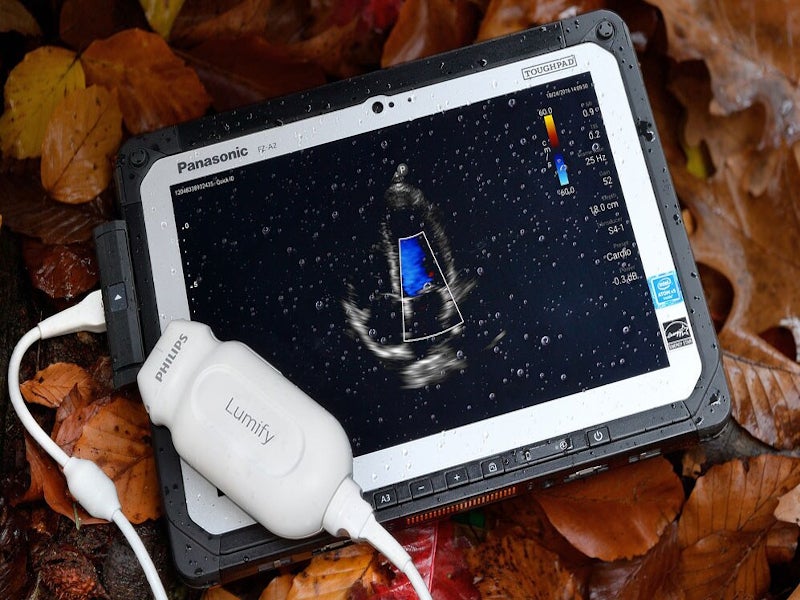
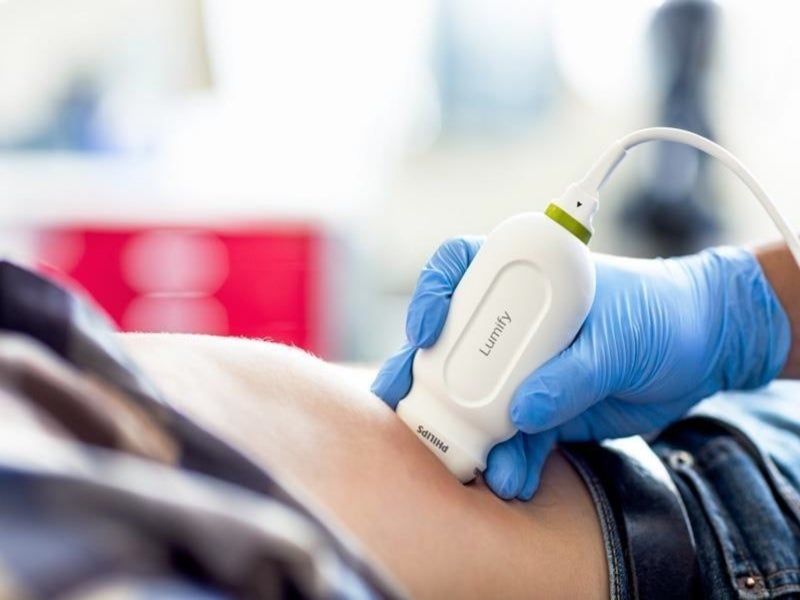
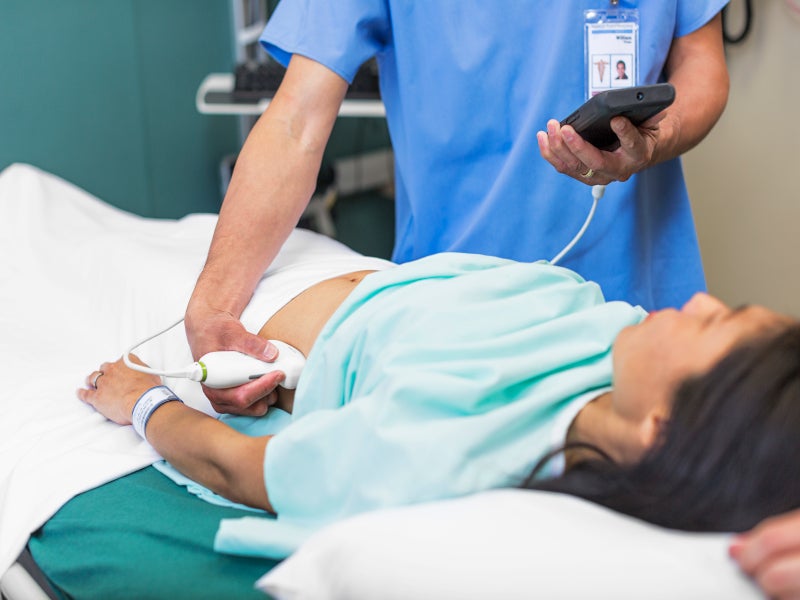
Lumify is a portable diagnostic ultrasound system developed by Philips for the ultrasound imaging and fluid flow analysis in various applications, including obstetric, cardiac, urology, cephalic, abdominal, cardiac foetal echo, gynaecological, musculoskeletal, paediatric, small organ and carotid.
Lumify received the 510(k) clearance from the US Food and Drug Administration (FDA) under the class II medical device category, in October 2015. Its new S4-1 cardiac transducer received the FDA approval in October 2016.
The FDA cleared Lumify with Reacts handheld tele-ultrasound solution for the management of lung and cardiac complications related to Covid-19 in May 2020. The device was launched in Japan in June 2020.
Lumify ultrasound system details
Lumify is a handheld, general-purpose, software-controlled ultrasound system, which integrates three USB transducers with centre line marker, namely C5-2 curved array, L12-4 linear array, and S4-1 sector array.
The system also comprises a commercial off-the-shelf (COTS) mobile device, Philips Ultrasound software and the Lumify Power Module (LPM). The ultrasound imaging is performed in B (two-dimensional), Colour Doppler, Combined (B+Color) and M modes.
It is compatible with smart devices such as Samsung Galaxy Tab S2 8in and can be used with mounting solutions.
Other accessories available with the system include quick-release replaceable cables, charging cables, rugged tablet and carry cases, ultrasound gel bottle of 0.25l, user manual, quick-start guide, Lumify carry bag and other rugged components for a specific demanding environment.
Lumify ultrasound system transducers details
S4-1 is broadband phased array transducer with the extended operating frequency range of 1-4MHz, used for the high-resolution imaging for abdominal and cardiac applications.
C5-2 is a broadband curved array transducer with the extended operating frequency range of 2-5MHz. The radius of curvature of the transducer is 50mm, used for the high-resolution imaging for deeper applications such as abdominal, gallbladder, obstetric or gynaecological and lung.
Lumify L12-4 broadband linear array transducer operates at a frequency range of 4-12MHz. With the aperture size of 34mm, the transducer provides high-resolution imaging for shallow applications including soft tissue, vascular, musculoskeletal and lung.
Operational characteristics of Lumify
Lumify system has an IP67 rating and can withstand dust and water to one metre for 30 minutes. It has been tested to withstand three feet drop to a hard surface, whereas the accessories in the carry case can withstand four feet drop to a hard surface.
The device can survive extreme temperatures ranging between -40oC and +71oC. It meets the MIL-STD-810G, IEC 60529, IEC 60601-1 and RTCA DO-160G standards.
Lumify ultrasound system for COVID-19 management
Ultrasound is helpful in imaging the peripheral lung tissues, which get affected by pneumonia, a Covid-19 lung symptom. The symptom increases the risk of cardiac complications. The cardiac ultrasound will allow evaluation of the effect of Covid-19 progression on the cardiac function.
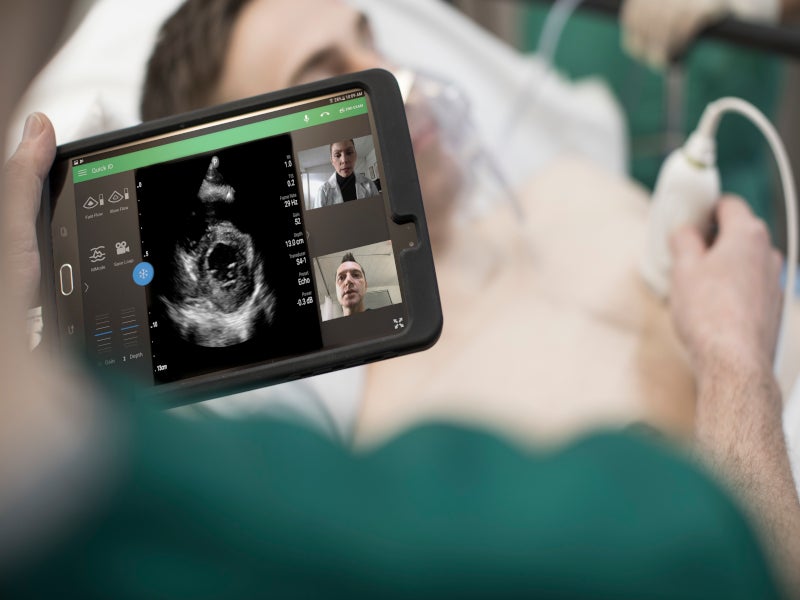
Powered by Reacts point-of-care tele-ultrasound solution, Lumify improves cardiac ultrasound imaging capabilities. It works with a compatible smartphone or tablet and allows to collaborate in real-time by two-way audio-visual calls along with the live ultrasound examination streaming of the patient.
Both healthcare professional and patient can simultaneously see the live ultrasound image and the positioning of the probe and discuss in real-time. The use of Reacts on Lumify minimises the transmission risk of the Coronavirus between patients and healthcare professionals.