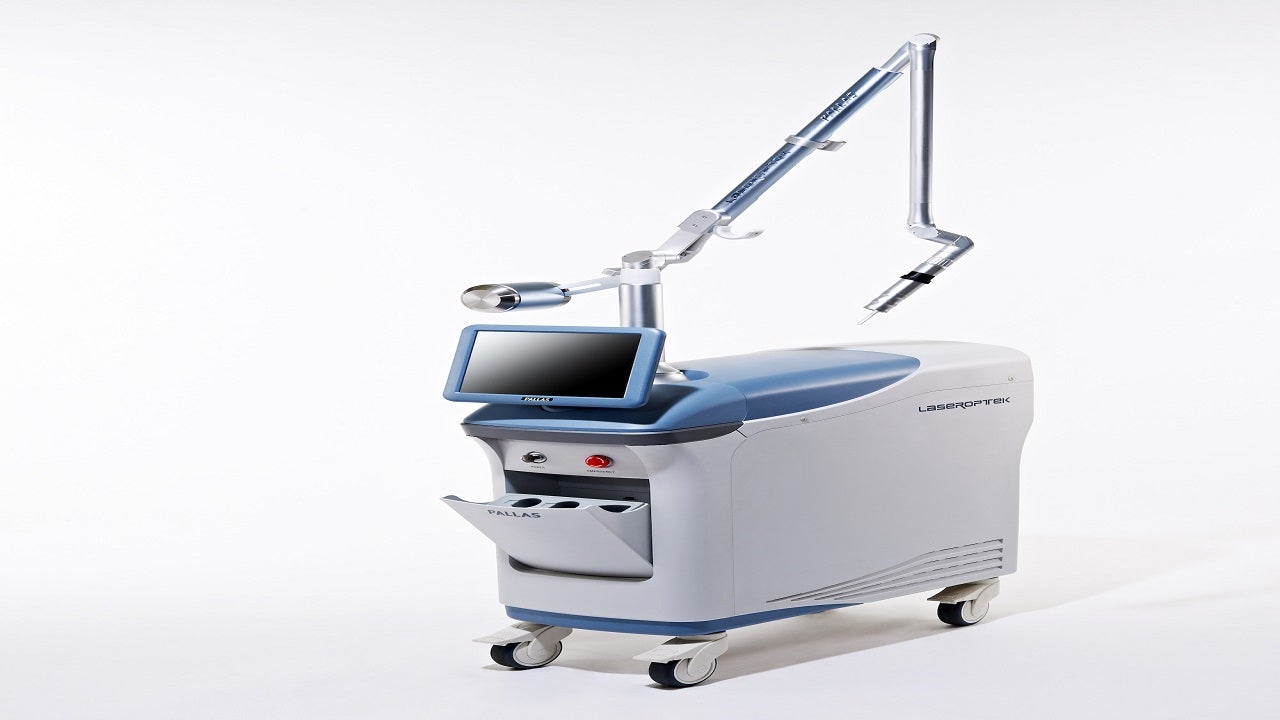
Manufactured by LASEROPTEK, a South Korean medical device company, PALLAS is the world’s first solid-state UVB laser device available in 308nm or 311nm versions.
The device is suitable for the phototherapy treatment of various dermatological disorders such as vitiligo, psoriasis, leukoderma, atopic dermatitis and alopecia areata.
LASEROPTEK partnered with Gale Force Aesthetics for the exclusive distribution of the device in the US. Gale Force will also provide installation and warranty services to the US customers through collaboration with Laser Service Solution.
PALLAS approvals
LASEROPTEK’s PALLAS was initially approved by the Ministry of Food and Drug Safety (MFDS), formerly the Korean Food and Drug Administration (KFDA) in 2015, and granted European CE mark in 2016.
The US Food and Drug Administration (FDA) cleared the device in 2017.
Details of PALLAS solid-state UV laser system
The PALLAS laser system is the world’s first gain-switched medical equipment with Ti: Sapphire solid-state 311nm UV-B laser.
It features an adjustable collimator handpiece with a wide spot size of 1mm to 12mm and ARM processor for electrical control. The device has a touch display with a 10.4in TFT LCD screen and closed-cycle water to air heat exchanger cooling system.
Its 311nm UV laser and stable output system deliver more effective treatment of psoriasis, vitiligo and eczema.
Narrow-band UVB wavelength is ideal for the treatment of various skin disorders such as psoriasis and vitiligo. The 311nm wavelength is closer to UVB wavelength, offering deeper penetration and eliminating undesirable burning effects.
This promising feature offers a new solution to innovative therapy that maximises the impact, safety and effectiveness of the laser treatment.
Benefits of PALLAS solid-state UV laser system
PALLAS solid-state laser system works with the optimal wavelength range of 311nm, which is the most stable and efficient wavelength for a UV system for treating dermatological conditions. It offers painless laser treatment to the customers.
Revolutionary 311nm laser system can penetrate deeper than the 308nm excimer lasers. PALLAS does not require tips or consumables, reducing the cost of maintenance and offering long-term usage, avoiding periodic servicing.
Solid-state technology eliminates the requirement of expensive and time-consuming excimer gas and electrode replacements, reducing laser downtime due to gas or electrode replacement services. The replacement of gas-based excimer lasers also improves laser operating efficiency. It eliminates the risk of gas exposure. Thus, it provides fully optimised and a user-friendly laser system.
Clinical study
A study shows that PALLAS Ti: Sapphire solid-state 311nm laser (TSL) based on narrow-band UVB illustrates similar efficacy and safety to 308nm excimer laser (EL) in the treatment of localised vitiligo.
In the study, 21 patients with 74 paired vitiligo lesions were randomised to either TSL or EL group. The lesions were treated twice a week for 12 weeks and examined every four weeks. The level of re-pigmentation in each group was measured and compared.
The TSL group showed a therapeutic effect similar to the EL group. Re-pigmentation results were excellent, particularly in 65% of facial lesions after 12 weeks of treatment with TSL therapy compared to 60% re-pigmentation in the EL group.
The results were promising as the laser therapies are commonly used for particularly, localised vitiligo and facial lesions treatment.