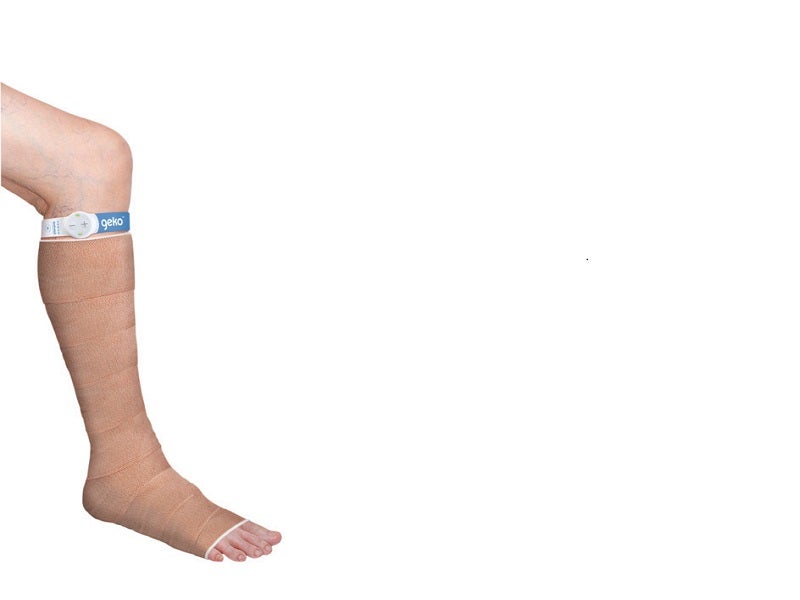
The geko™ device is an easy-to-use, disposable, wearable neuromuscular electrostimulation (NMES) device designed to enhance blood flow in the deep veins of the leg, preventing venous thromboembolism (VTE) and reducing oedema.
Developed by the UK-based medical device company Sky Medical Technology, geko works without applying any external pressure on the leg and facilitates complete movement.
Firstkind, a subsidiary of Sky Medical Technology, manufactures the device.
The device was launched at a major orthopaedic conference in the US in January 2020.
Patients with hard-to-heal venous leg ulcers showed a higher healing rate with the geko device compared to standard-of-care alone in a landmark multicentre randomised self-controlled trial in 2023.
Regulatory approvals for geko device
The geko device received 510(k) approval from the US Food and Drug Administration (FDA) for improving microcirculatory blood flow in the lower limb soft tissue of patients with venous insufficiency or ischemia, in December 2021.
The approval was extended to the new (W3) geko device variant for the same condition in March 2022.
The device also holds the European CE mark under the class IIa category to improve blood circulation, prevent blood clots and promote wound healing.
The geko device was made available on the Government of India e-marketplace procurement portal in April 2022.
The portal enables government hospitals in India to procure the device for use in preventing blood clots, managing post-surgical swelling in orthopaedic patients, and aiding the healing of chronic wounds such as leg ulcers.
The device was adopted by the Internal Medicine Clinic at the Al Mahmoud Medical Center in Doha, Qatar, in December 2022.
The device is exclusively distributed by Al Raya Medical Equipment in Qatar.
In March 2023, the device received certification for compliance with the UK Conformity Assessed Mark, a regulatory mark required for medical devices sold in the UK.
In November 2023, the geko device received reimbursement in the UK, allowing clinicians to propose its inclusion in the local Integrated Care System formulary to the relevant approval committee.
The device was made available through the UK National Health Service Supply Chain in May 2024.
geko device details
The geko is a portable device embedded with OnPulse™ NMES technology, resembling a small wristwatch.
It is a one-size-fits-all, self-adhesive device worn on the knee to prevent the need for more complex procedures.
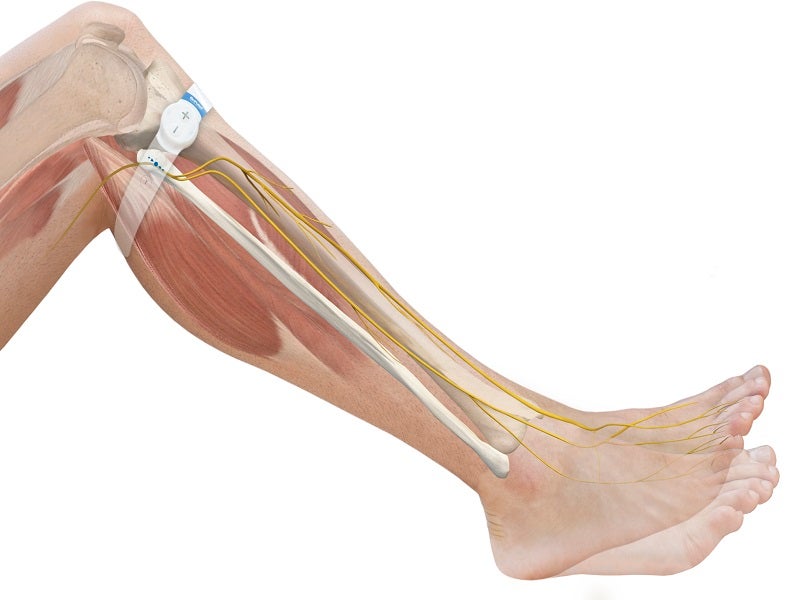
The device sends small electrical impulses that gently stimulate the common peroneal nerve. The stimulation activates the calf and foot muscle pumps, resulting in enhanced blood flow in the deep veins of the calf.
Clinically, the device has been shown to enhance blood flow, aiding in the prevention and treatment of various acute medical disorders by stimulating the body’s natural nerve impulses to increase circulation.
The neuromuscular stimulation in the lower leg mimics the normal walking process up to 60% of the time, without requiring movement or exertion, and causes no discomfort to the patient.
geko device features and benefits
Weighing just 10g, geko sends 60 pulses per minute and offers ten stimulation settings to accommodate a range of patient responses to stimulation.
It comes without cables or leads, creates no tripping hazards, and allows patients to move freely.
It is safe and hygienic with a battery life of up to 24 hours.
The device provides painless 1Hz muscle contractions per second, increasing superficial femoral venous volume flow by 100%, femoral arterial volume flow by 75%, and microcirculatory flux to the dorsum of the foot and thigh by 400%.
The new (W3) geko device offers the added advantage of a third electrode, which enhances nerve stimulation, improves patient comfort, and leads to better patient outcomes.
Additionally, the device provides two 12-hour therapeutic doses, doubling the duration compared to the previous (W2) device, which offered two six-hour doses.
OnPulse technology
The clinically proven OnPulse technology integrated into the geko device combines neuromuscular electrostimulation with an easy-to-use wearable design.
During low-frequency nerve stimulation, OnPulse delivers the current transcutaneously by non-invasive skin surface electrodes placed on the common peroneal nerve located around the fibula’s neck, behind the fibula’s head.
The conductive hydrogel is used for adhesion and current flow from the electrode into the skin.
The technology activates the motor neurons within the nerve bundle, which send impulses to the muscle fibres, causing them to contract.
OnPulse stimulates the tibialis, peroneus longus, and lateral gastrocnemius muscles simultaneously by stimulating the same peroneal nerve around the posterior or anterior bifurcation.
The synchronous contraction compresses the venous system, emptying blood in the deep veins of the calf at a rate equivalent to 60% of walking, with no change in heart rate or blood pressure.
OnPulse differs from traditional NMES devices such as muscle stimulators, which apply direct electrical stimulation to the muscle mass to force contraction inside a single muscle, sometimes utilising huge and uncomfortable stimulus amplitudes.