 On 29 September, Abbott received FDA approval for the FreeStyle Libre Flash continuous glucose monitor (CGM). Though the device received CE mark approval in 2014, it the first FDA-approved CGM to not require the standard finger pricking for system calibration, signifying a milestone in diabetes care and management for US patients.
On 29 September, Abbott received FDA approval for the FreeStyle Libre Flash continuous glucose monitor (CGM). Though the device received CE mark approval in 2014, it the first FDA-approved CGM to not require the standard finger pricking for system calibration, signifying a milestone in diabetes care and management for US patients.
The FreeStyle Libre sensor can be worn for up to 10 days at time, during which time a hand held reader can be used to capture and display blood glucose data. The continuous data stream will serve to fill in gaps that exist with traditional blood glucose monitoring systems, and make physician and patient management of diet, treatment, and lifestyle more convenient.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
GlobalData estimates the global market for blood glucose monitors, comprising CGMs and traditional glucose meters, was valued at $1.6B in 2017. Driven by the expanding diabetic population and the need for remote, patient self-monitoring systems, the market is expected to grow at a Compound Annual Growth Rate (CAGR) of 7.7% through 2022. CGMs accounted for nearly 48% of revenue in 2017, and are expected to grow rapidly at a CAGR of 10.3% to surpass traditional glucose meters’ revenue by 2020.
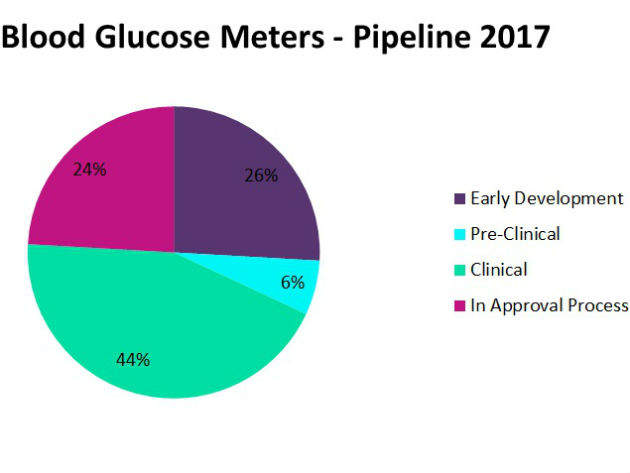
GlobalData anticipates increasing competition in the glucose monitoring market, as innovative solutions integrating CGM and wireless capabilities emerge. Bolstered by the need for improved systems, the glucose monitoring pipeline is bursting with nearly 200 blood glucose meters, both traditional and CGM, in various stages of development. Nearly half of these systems are in the clinical phase, as the race to market the most advanced glucose monitoring system intensifies.





