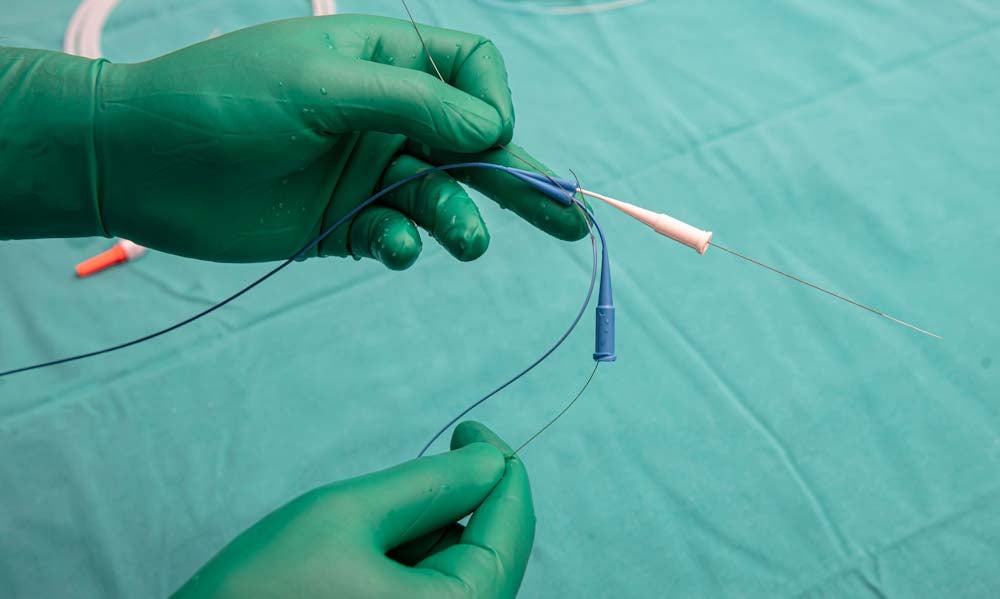
Guidewires are an essential component of various diagnostic and therapeutic medical procedures for vascular therapy. They are flexible, precisely controlled wires that are used to access the coronary and peripheral vascular system and are commonly used for placing and guiding devices. These procedures are a minimally invasive technique for accessing blood vessels.
The European guidewire market has seen significant growth in recent years, and this trajectory is expected to continue. In fact, the market is predicted to grow at a CAGR of 5.08% between 2022 to 2027. Two factors may be contributing to this growth; the increase in cardiovascular disease and the expanding trend towards minimally invasive surgery.
According to a GlobalData report on the coronary guidewires market size between 2015-2030, cardiovascular disease is the leading cause of death across Europe. The steady growth of the coronary guidewire market is due to the growth of percutaneous coronary intervention (PCI) procedures in the European region.
Similarly, for the peripheral guidewires market, GlobalData states that Europe is a key region of growth. Globally, the increase in the ageing population is one of the main driving factors, as it means that peripheral vascular disease or peripheral artery disease (PAD) is of growing concern.
In addition to an ageing population, the European Heart Network suggests in its cardiovascular disease statistics that dietary factors make the largest contribution of all behavioural risk factors. High systolic blood pressure makes the largest contribution of all the medical risk factors. Additionally, while smoking rates have decreased across much of Europe, the pace of decline has slowed and rates remain stable or are rising in some countries, particularly among women.
The minimally invasive surgical instruments market overall is expected to grow, especially as innovations in surgical robots continue. The growing demand for these types of surgeries, rather than more invasive heart surgery, is due to quicker recovery, smaller incisions, reduced scarring and pain, and shorter hospital stays.
To make them suitable for these surgeries, guidewires must offer excellent biocompatibility, moderate tensile strength, and good corrosion resistance, as well as moderate fatigue and relaxation resistance. They also must be flexible and have coatings that offer the lubricity to easily manoeuvre around the body.
Under its Exera® brand, Alleima (formerly Sandvik Materials Technology) has designed and developed wire for guidewires across the globe. Recently, through the acquisition of Polyfil (experts in fine-wire manufacturing) and Galvarex (experts in surface treatments and electroplating), Alleima has expanded its production capabilities in Europe.
Materials that have proven to be effective for guidewires include austenitic stainless steels Exera® 12R10 HV (moderate tensile strength, good corrosion resistance, moderate fatigue and relaxation resistance) and molybdenum-alloyed high-strength vacuum-remelted Exera® 316LVM (excellent micro-cleanliness and structural homogeneity) medical wire. Exera® cored medical wire (SBCW) is also used, which is a composite wire that combines Exera® F562 medical wire CoCrMo (MP35N) alloy shell and a secondary alloy as the core material to achieve excellent corrosion and fatigue resistance.
In order to reduce friction, coronary guidewires are coated with lubricious materials. Alleima offers a range of surface treatments, including Polyimide and PTFE coatings with thickness ranges from 0.00127mm to 0.0127mm.