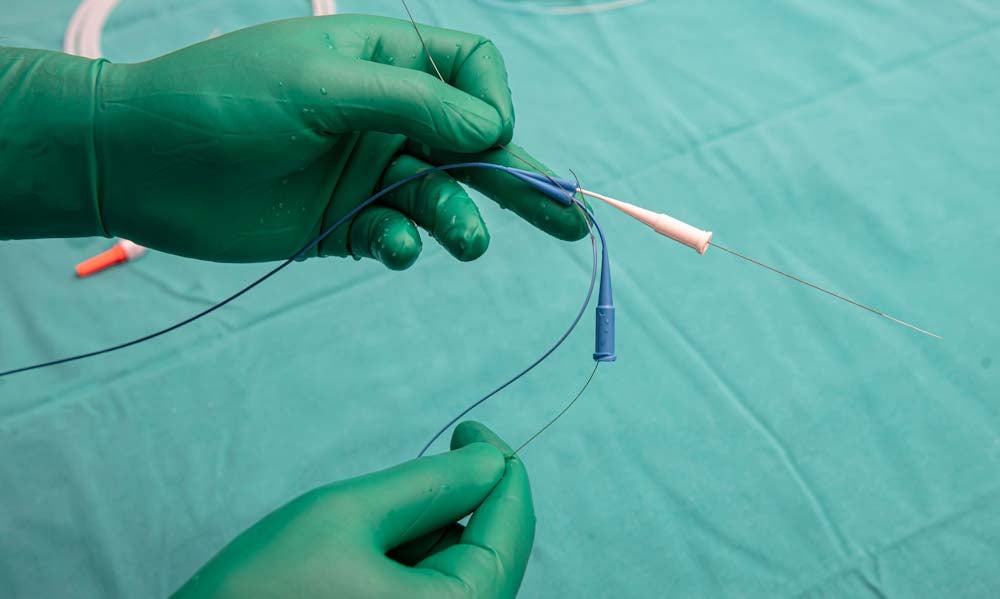
Industry 4.0 has launched an era of medical devices that are smaller and smarter. Not only are these devices providing healthcare professionals with more accurate real-time data for improved diagnoses and treatments, but the impact on patients’ quality of life is minimised.
A patient’s quality of life can be improved by medical devices in a number of ways, and one significant factor is that hospital visits can become shorter and less frequent. With remote monitoring, patients can self-monitor and provide healthcare professionals with important data without needing to make an appointment.
According to GlobalData market insight, the remote patient monitoring (RPM) market is expected to double over the next five years, citing the Covid-19 pandemic as a catalyst for market growth. Patients that rely on medical devices are more likely to be at risk, especially among the elderly or those with autoimmune disorders. In addition to the hospital visit itself, remote monitoring means that patients are not required to travel frequently, taking the pressure off elderly or disabled patients who may find this difficult, as well as simply being more convenient.
One commonly used medical device is the Continuous Glucose Monitor (CGM). This means that diabetes patients no longer need to carry out manual finger-prick tests or wait until symptoms show. This also means that should a patient’s condition become critical, healthcare providers or friends and family can be immediately alerted.
It is not just the frequency of hospital visits that is reduced, but also the length of the stay itself. Preventative action enabled by continuous monitoring reduces the risk of a patient’s condition escalating to the point where long stays are required, and catheters and guidewires mean that surgeons can identify problems or implant devices without needing to perform invasive surgery. Alongside the rise in remote monitoring, the minimally invasive surgery (MIS) market is also expected to grow.
Advances in medical wire are making all of this possible, from guidewires that reduce the need for invasive surgery to wires in heart monitoring devices that sense irregularities even before external symptoms occur. With the demand for non-invasive implantable devices and remote monitoring capabilities means that medical device manufacturers need much finer wire that still has the appropriate strength and fatigue resistance, as well as requiring much more advanced sensing and transmitting capabilities.
With medical wire becoming more complex in terms of the required capabilities (such as flexibility, conductivity, and biocompatibility), as well as needing to be much smaller and more precise, many manufacturers do not have the in-house expertise. Manufacturers are increasingly outsourcing medical wire components and looking for a supplier that can assist in design and production, from choosing the right materials to expertise in wire grinding and medical-grade coatings.
Alleima, formerly Sandvik Materials Technology, supplies a broad range of ultra-fine medical wire under its EXERA® brand, and its capabilities in wire insulations and surface treatments are continuing to grow through its acquisition of Polyfil and Galvarex. EXERA® medical wire is used in medical devices to transmit, sense or stimulate within the human body, utilising expertise in metallurgy, more than 200 alloys, and various customisations such as coiling and coatings.
With these exciting advancements on the horizon, now is the ideal time to form partnerships with medical device component experts to work together and design a product that can change patients’ lives. For more information about how Alleima is providing vital components for remote monitoring devices, download the whitepaper below.