Vertellus Biomaterials
Biocompatible Coating Materials
Vertellus Biomaterials is a business unit of Vertellus Specialty Materials and offers a range of clinically proven biocompatible materials known collectively as PC Technology™.
Subscribed
You have successfully submitted your enquiry. Someone from our company will respond ASAP
About Us
Vertellus Biomaterials is a business unit of Vertellus Specialty Materials and offers a range of clinically proven biocompatible materials known collectively as PC Technology™.
PC Technology comprises monomers and polymers incorporating phosphorylcholine (PC) for use as coatings to improve the biocompatibility of medical devices or as bulk materials used in the manufacture of contact lenses. Gels and solution products can also be made.
PC materials have antifouling properties and are used to improve blood compatibility, and reduce inflammatory response and biofilm formation. These products have proven drug delivery capability.
Vertellus Biomaterials develops coating processes for customers and offers both off-the-shelf and bespoke biomaterials.
Biocompatible coating materials to prevent protein fouling
Phosphorylcholine (PC) is the predominant head group found in phospholipids that form the bi-layers making up cell membranes. PC is zwitterionic; it contains both positive and negative charges but is overall electrically neutral. Being highly polar the PC group is hydrated and this tightly held water layer makes it energetically unfavourable for proteins and cells to irreversibly bind to a surface incorporating PC.
By virtue of their anti-fouling nature, PC materials have been shown to improve the biocompatibility and performance of medical devices through reduced:
- Protein deposition and activation1
- Blood activation and thrombus formation2, 3
- Bacterial adhesion and biofilm deposition4
- Inflammatory response5
- Fibrous capsule formation6
PC materials can be formulated to produce lubricious, instantly wettable surfaces and have been used for the controlled local delivery of drugs, including anti-inflammatories, anti-proliferatives and antibacterial agents.
PC coatings on medical devices show improved haemocompatibility. PC coated heart-lung machines reduce platelet activation and lower blood loss during cardiopulmonary bypass surgery. PC-coated coronary guide-wires aid in the prevention of thrombus formation on the guide-wire tip during short-term clinical use (FDA-allowed claim)2.
Reduced biofilm formation
Materials coated with PC have been shown to suppress bacterial adhesion and biofilm formation3, 5.
Biocompatible polymers for controlled drug delivery
PC-polymers are typically applied using a simple dip or spray coating process using organic or aqueous solvent systems. This allows for application of a suitably soluble drug by applying a mixture of the active with the coating, or simply swelling the coating in a solution of the drug after application. Release rates may be controlled by modifying the chemical structure of the polymer to alter interactions with the drug, through the use of a top-coat barrier or through post coating curing to introduce cross-linking and tailoring the water content to provide the desired elution profile.
Publications on PC coated drug-eluting coronary stents include work on angiopeptin, dexamethasone and methyl prednisolone, with the latter demonstrating reductions in acute inflammatory reactions. Zotralimus, 17ß-estradiol and the matrix metalloproteinase inhibitor, batimastat have been studied and gene therapy has also been successfully delivered from PC coatings.
PC coated drug eluting products that have been commercialised include Abbott’s Dexamet™ stent incorporating dexamethasone and Medtronic’s Endeavor® zotralimus eluting stent system.
PC polymers can be used as drug eluting coatings on other medical devices to deliver antimicrobials or anti-inflammatories, for example. Bulk polymers incorporating PC may be used to fabricate implantable depots with the potential advantage of reduced inflammatory response.
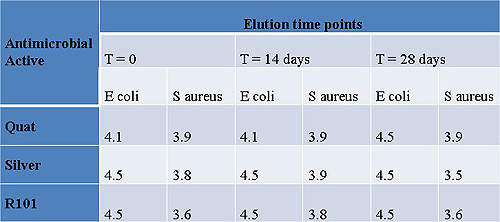
Antimicrobial coatings
Work evaluating PC materials in combination with active antimicrobials has demonstrated the controlled release of a number of agents over several weeks.
Contact lens materials
PC Technology has been successfully commercialised as a contact lens material by CooperVision in its Proclear range of products. Proclear has been shown to exhibit less on-eye dehydration and is less prone to tear film deposition. These properties contribute to its ability to maintain a stable tear-film which results in improved comfort. Proclear is the only contact lens with an FDA allowed claim related to improved comfort.
Vertellus Biomaterials is developing a range of new PC materials which can be used in conjunction with siloxanes to provide the benefits of good wettability and gas permeability.
PC Technology – proven biomaterials
The PC Technology is licensed to a number of leading medical device manufacturers, including CooperVision, Medtronic, Abbott and the Sorin Group. There are numerous FDA Master Files relating to the materials and device incorporating them.
Vertellus Biomaterials looks forward to working with medical device companies interested in improving the clinical performance of their products.
References
- Lewis, A. L., Colloids Surf., B Biointerfaces 2000, 18, 261-275
- Gobeil, F., et al., Can J Cardiol., 2002, 18(3), 263-9
- Ranucci, M., et al., Int.J.Artif.Organs., 2004, 27 (4), 311-9
- Berry, J.A, et al., Otolaryngol Head Neck Surg, 2000,123, 246-51
- Goreish, H.H., et al., J Biomed Mater Res A, 2004, 68A, 1-9
- Link, C., et al., Plastic and Reconstructive Surgery, 2011, 128,1, 22-3.
Contact Details
Website
Email Address
Address
Herriard,
Hampshire,
Basingstoke,
RG25 2PH,
Other,
United Kingdom