Scientists develop intelligent surgical knife to detect cancerous tissue
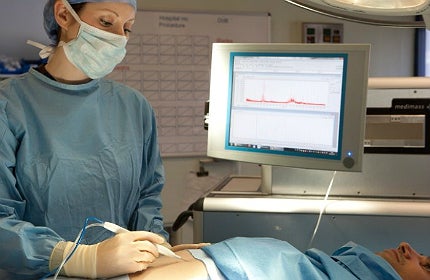
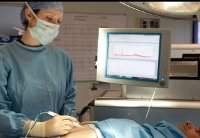
Researchers at Imperial College London in the UK have created an intelligent knife, which can help surgeons identify cancerous tissue.
The iKnife is based on an electrosurgery technology invented in the 1920s and widely used in the medical field today.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
With the help of an electrical current, an electrosurgical knife can quickly heat the tissue and then cut through it, while simultaneously reducing the loss of blood.
Abbott to expand medical device business with two acquisitions

Abbott is expanding its medical device business with the $710m acquisition of OptiMedica, a Silicon Valley-based ophthalmic device firm, and IDEV Technologies, a Texas-based stent manufacturer.
Under the agreement, Abbott will acquire OptiMedica for $250m in an all-cash deal.
In addition, the firm will make payments of up to $150m following achievement of certain sales, regulatory or development milestones.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataBD gets approval for integrated IV catheter system for subcutaneous therapy

Global medical device maker Becton, Dickinson and Company (BD) has received approval from the European regulatory authorities for its BD Saf-T-Intima Safety Integrated IV Catheter System, which can be used in subcutaneous infusion therapies.
Made with BD Vialon biomaterial, the system’s pre-attached extension line lessens the cost in assembly time and also the risk of contamination.
BD claimed the increase in-dwell times reduces overall costs of the therapy used besides catheter materials.
Physio-Control’s latest chest compression system provides better CPR

Physio-Control has developed Lucas 2, a system that delivers continuous chest compressions in order to maintain blood flow to the brain and other organs when a person suffers a cardiac arrest.
The latest chest compression system provides better cardio-pulmonary resuscitation (CPR) as the treatment in cardiac arrest cases should begin within four to six minutes, before the heart and brain are damaged.
CPR is an emergency procedure to keep the brain function intact until additional measures are taken to restore blood flow and breathing for a person suffering a heart attack.
Techne closes acquisition of haematology products maker Bionostics
Minneapolis-based Techne has closed the acquisition of Bionostics Holdings and its subsidiary Bionostics in a cash deal worth $104m.
Techne announced its plans to acquire Bionostics and the operating subsidiary last month, as part of its strategy to boost its haematology products portfolio.
Techne CEO and president Charles R Kummeth said the acquisition of Bionostics would add capabilities in new areas such as coagulation and expands its controls portfolio, giving the company the critical mass it needs to remain competitive and offer its customers continued value and options to serve their needs.
Mindray Medical closes acquisition of Zonare Medical Systems
China-based medical devices developer Mindray Medical International has completed the acquisition of Zonare Medical Systems, a US-based ultrasound technology provider, for $101.7m, as part of its strategy to boost its presence in the ultrasound market, particularly in the US.
Mindray entered into a definitive agreement to acquire Zonare on 12 June 2013.
The company then announced that it would fund the acquisition with existing cash and third-party borrowings, and expects the deal to be slightly dilutive to its full-year 2013 and 2014 earnings.
Elekta’s Clarity 4D Monitoring system for prostate cancer secures CE Mark
Sweden-based Elekta has secured the CE Mark for its Clarity 4D Monitoring system, which will allow doctors to monitor the motion of prostate with an accuracy of even sub-millimetres during radiotherapy.
During advanced prostate protocols, such as reduced margin hypofractionated therapy or stereotactic ablative body radiotherapy (SABR), it is important for doctors to keep monitoring not only the prostate but also the surrounding anatomy, such as bladder, rectum and penile bulb.
Usually, doctors avoid the surrounding anatomy in order to reduce the treatment side effects such as erectile dysfunction, incontinence or rectal bleeding.
Given Imaging’s PillCam COLON video capsule receives regulatory clearance
Given Imaging has received clearance from the Pharmaceuticals & Medical Devices Agency (PMDA) of Japan for its PillCam COLON, a video capsule to diagnose colonic disease in patients unable to have a colonoscopy.
The 11mm X 31mm PillCam COLON video capsule features two miniature colour video cameras on both sides, a battery and an LED light source.
Once ingested, it can transmit up to 35 frames per second for approximately ten hours to a recording device worn by the patient.
The data obtained from the capsule is transferred to a computer using RAPID software, with the video data then compiled, allowing doctors to review and report the results of the PillCam study.