ML Electronics
Medical Electronics Product Design and Development
Subscribed
You have successfully submitted your enquiry. Someone from our company will respond ASAP
About Us
ML Electronics (MLE) offers specialist medical electronics design and product development. ML Electronics is an ISO 13485 certified services company specialising in embedded electronics, user interfaces and software for medical applications.
Clients’ products include medical device electronics; healthcare systems; and assisted living, well-being and fitness, home and clinic products.
Medical device electronics
Specialist technical areas include analogue design, instrumentation and control, real-time data and signal processing, system-on-chip (SoC), advanced embedded processing, 16bit and 32bit microprocessors, DSP, FPGA, analogue power supply design and techniques using the latest digital power supply technology.
Custom medical electronics product design and manufacture
ML Electronics offers a complete range of services from concept through to production. The philosophy at ML Electronics is to use design as a tool for commercial success. This is achieved by combining customer insights and commercial awareness with innovative thinking and technical knowledge to design and produce differentiating products. This ensures ML Electronics assists its clients to develop innovative and practical products that sell in their marketplace.
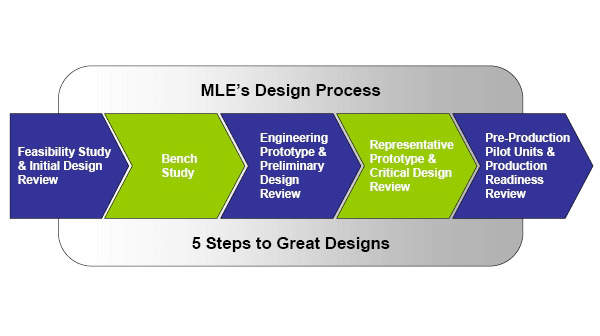
Medical design methodology
At ML Electronics our unique five-stage development process means that we can develop cost-effective turnkey systems from initial feasibility studies to full production manufacture.
Our design and manufacturing processes are approved and audited to ISO 9001 and we have achieved Scope extension to this quality standard to enable us to specifically design software. This process is audited by Tick-IT inspectors. We have a very broad experience of various quality standards and processes required for different marketplaces, including supporting clients in achieving FDA approval.
Medical electronics verification validation with FDA
ML Electronics has proven industry experience in medical device development that utilises FMEA techniques and risk analysis for built-in reliability and cost effectiveness that fulfils your market needs. With experience in designs to meet IEC 60601-1, ML Electronics ensures that we deliver products to the highest medical standards. We have many years of experience with verification validation testing and in FDA regulations and standards.
Medical electronics product manufacture
With production facilities, ML Electronics can provide technology demonstrators, prototypes and production units fully tested to your requirements. For high volume manufacture we can provide support with your chosen manufacturing facility.
Contact Details
Website
Email Address
Address
Whiteparish,
Wiltshire,
Salisbury,
SP5 2QE,
Other,
United Kingdom