The Use of Next Generation Nitinol for Medical Implants
This article reviews the use of Nitinol endovascular devices and the effects of biomechanics, design, and Nitinol microstrucutural purity on fracture.

EUROFLEX is a leading global manufacturer of high-quality semi-finished products and components for medical devices.
You have successfully submitted your enquiry. Someone from our company will respond ASAP

EUROFLEX is a leading global manufacturer of high-quality semi-finished products and components for medical devices.
As a leading specialist for solutions made from self-expandable Nitinol, balloon-expandable cobalt-chromium and stainless-steel alloys and other innovative biocompatible materials, EUROFLEX is the competent partner for numerous companies in the field of medical technology.
Certified under the German Institute for Standardisation (DIN) European Standards (EN) International Organisation of Standardisation (ISO) 9001 and DIN EN ISO 13485, the management system ensures a consistently high level of product quality.
Nitinol, cobalt-chromium and stainless-steel products supplied by EUROFLEX are used worldwide in countless US Food and Drug Administration (FDA), China Food and Drug Administration (CFDA) and European Conformity (CE) approved implants and devices such as coronary, peripheral and retriever stents, heart valve frames and filters.
With manufacturing facilities in Germany and Costa Rica, as well as a standard-setting testing laboratory in the US, EUROFLEX delivers its alloys, components and service worldwide.
EUROFLEX specialises in processing and distributing metallic biocompatible alloys and components with stringent tolerances for the medical technology sector. These are available as seamless high precision tubes, metal compounds, wires or sheets.
Typically, EUROFLEX processes mill products into semi-finished products with a focus on tight tolerances and surface finish.
The company’s core business is the processing of Nitinol (nickel-titanium) ((American Society for Testing and Materials) ASTM F2063), cobalt-chromium (L605 according to ASTM F90, MP35N according to ASTM F562) and stainless-steel tubes (316L according to ASTM F138, Ni-free stainless, 304) for implants and other high-quality finished devices.
EUROFLEX manufactures Nitinol tubes to client specifications, adapted to customer post-process capabilities. Depending on laser technologies, shape-setting and surface processes, the company can create tubing in a variety of surface conditions.
The company offers oxide outer and inner surfaces as mechanical (ground, polished), slurry cleaned and sand-blasted outer and inner surfaces.
Standard size range for the outer diameter is 0.06mm-20mm with tight tolerances for implants and wider tolerances for catheters. The customer can choose from five different Nitinol material grades from three Nitinol generations.
Over the years, EUROFLEX expanded its capabilities into materials such as titanium alloys (Ti-Grade 2, Grade23, beta-titanium alloys), platinum and bio-absorbable alloys for medical applications.
New minimally invasive treatment methods require new material concepts. For example, it should be possible to insert complex structures such as aortic or mitral valves through the femoral artery or the apex of the heart in a folded-up shape while simultaneously exhibiting a high fatigue resistance in a fully expanded state.
To meet these and similar requirements, EUROFLEX developed an ultrafine Nitinol, Nitinol HCF-SE (High-Cycle Fatigue SuperElastic condition), which, compared with standard versions, exhibits improved fatigue behaviour by a factor of one to two days.
Combined with materials such as tantalum or platinum alloys, it allows for the production of optimised materials that lead to new products and ultimately benefits the patient.
The company’s HCF-SE-wire combines expanded fatigue life with state-of-the-art reel-to-reel surface processing capabilities, delivered with an etched or electro-polished surface on spools.
EUROFLEX offers an extensive range of tube and sheet sizes from various alloys from stock. Sample quantities can be shipped within a few days to support the customer in the development phase, and credit card payments will soon be available.
Furthermore, the company has a rapid-response department with dedicated staff and machines, which can make custom runs within a few weeks, having access to vast material stock. The customer can submit sizing and other specifications or use the extensive experience from the company for the recommendation of a sufficient material specification.
For more complex developments and new alloys, EUROFLEX and it mother company G RAU, with its research and development staff, can manage large development projects to support customer concepts with next-generation alloys, processes and components.
The company’s test labs in Germany and the US can support customer developments with material testing such as tensile, compression and hardness tests, with corrosion and galvanic testing using ASTM standards and with fatigue testing using state-of-the-art test protocols. With dedicated experts, the support can continue till submission and beyond.
In addition to semi-finished products such as tubes, sheets and compound wires, EUROFLEX offers customers its expanded lines of medical components and new, custom-designed solutions.
EUROFLEX offers a wide range of manufacturing processes such as stamping, deep-drawing, cold forging and surface treatment options such as electro-polishing and plating, to create unique components with features that include shape-forming, turned and milled parts, and laser-processed parts.
The company tailors its manufacturing processes to customer needs, with quality and high precision always a top priority.
As a subsidiary company of G RAU with its headquarters in Pforzheim and great successes with its technologies in metal forming since 1877, EUROFLEX has the necessary experience to act as a development partner for our customers from the first design to high volume serial production.

This article reviews the use of Nitinol endovascular devices and the effects of biomechanics, design, and Nitinol microstrucutural purity on fracture.

As part of a multi-stage plan, we already started to expand the capacity in our Rapid Response department by 30% in the second half of 2020 by investing in new production equipment.

It is one of the main challenges found in medical devices using “traditional” materials like stainless steel i.e. in a handle for a delivery system for an implant that it will require an increasing amount of force the more it is deformed.
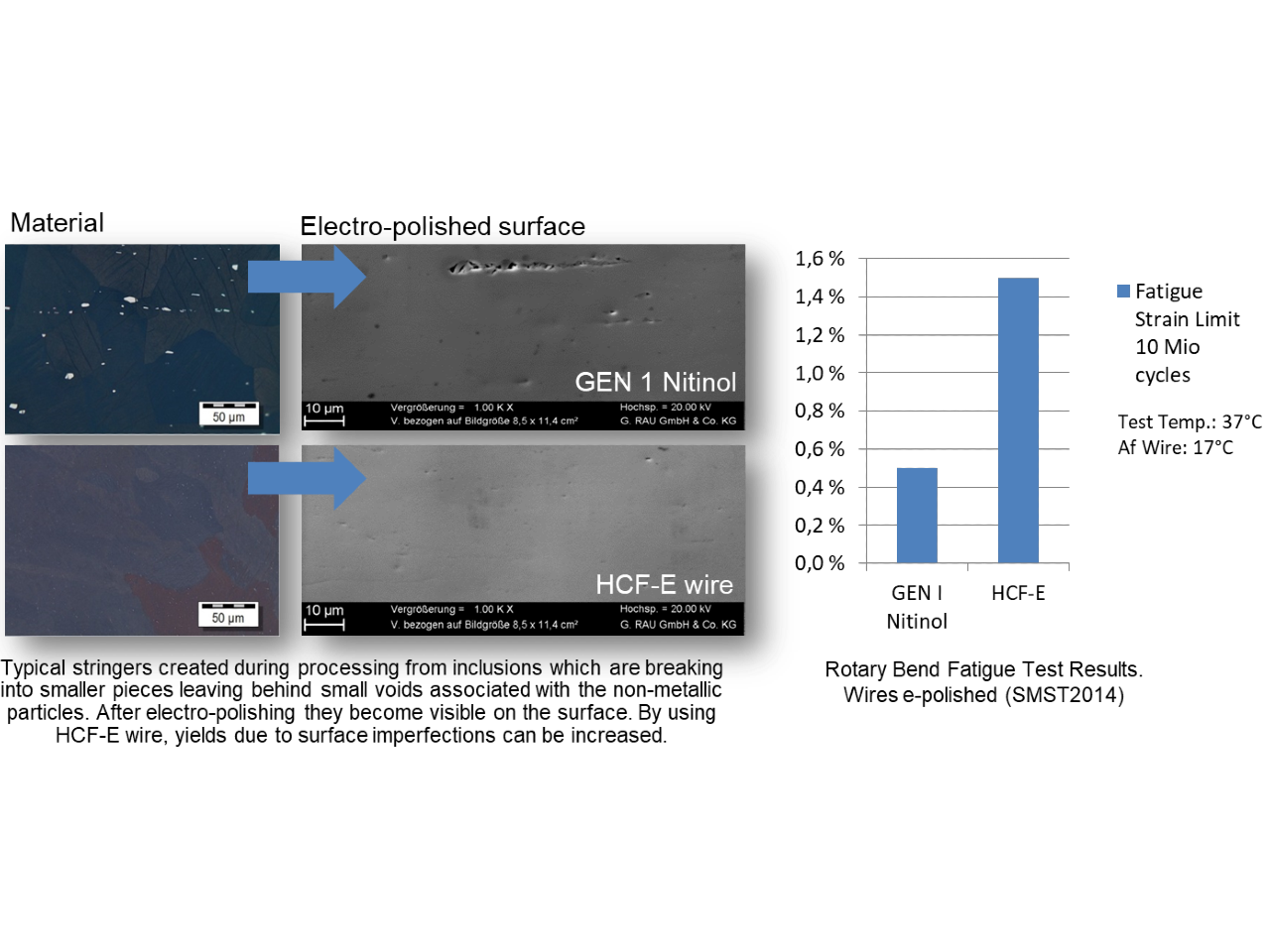
EUROFLEX has released electro-polished third-generation Nitinol HCF wires.

EUROFLEX is proud to announce the start of our tantalum tube manufacturing line. High-quality extra small tantalum tubes of customisable dimensions and proportions with diameters of min. 0,5 mm can now be produced.