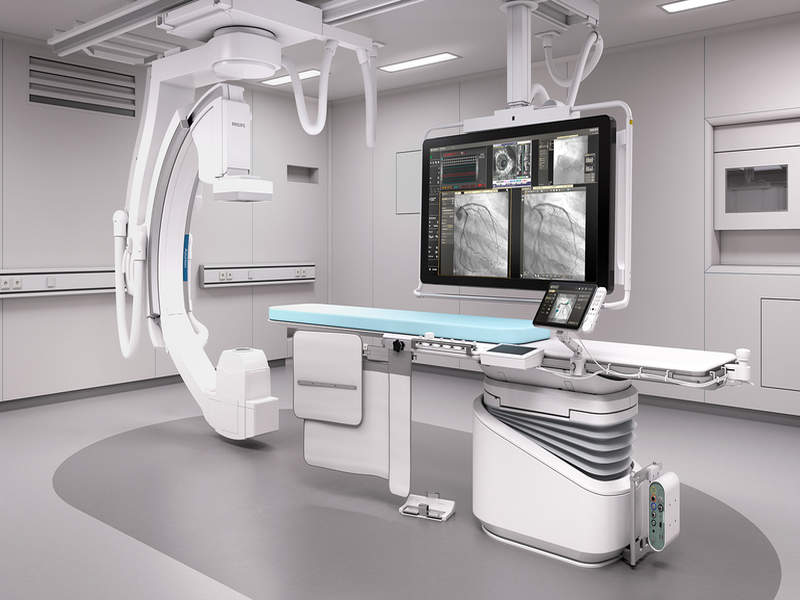
Philips launches new image-guided therapy platform Azurion
Netherlands-based health technology company Royal Philips expanded its existing portfolio of solutions with the launch of new image-guided therapy platform Azurion.
Meant for use in the interventional labs, Azurion features an ergonomic design with an easy-to-use user interface and is equipped with new workflow options, performance dashboards, as well as a range of productivity improvement services.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The platform includes 1,000 new components such as an improved flat-panel detector and the firm’s new ConnectOS operating system, which enable integration of real-time information from all relevant technologies in the lab.
Fujifilm gets FDA premarket approval for Digital Breast Tomosynthesis
Fujifilm Medical Systems USA secured premarket approval (PMA) from the US Food and Drug Administration (FDA) for its Digital Breast Tomosynthesis (DBT) option of ASPIRE Cristalle system.
DBT has been developed as an optional software upgrade for the digital mammography ASPIRE Cristalle FFDM system.
Designed to optimise patient dose and improve image quality, the ASPIRE Cristalle FFDM system with DBT utilises a combination of hexagonal close pattern (HCP) detector design, advanced image processing, and image acquisition workflow.
Philips and PathAI partner to develop AI diagnostic solutions
Health technology firm Royal Philips partnered with artificial intelligence (AI) technology company PathAI to develop diagnostic solutions for diseases such as cancer.
The firms will work towards applying the AI to massive pathology datasets by building deep learning applications in computational pathology for better diagnostic and treatment decisions.
Applications for automatic detection and quantification of cancerous lesions in breast cancer tissue will be developed initially.
Roche launches new prostate cancer diagnostic test
Swiss diagnostic firm Roche launched a new test called anti-p504s (SP116) Rabbit Monoclonal Primary Antibody for the diagnosis of prostate cancer.
Intended for the use in laboratories, the anti-p504s (SP116) Rabbit Monoclonal Primary Antibody is designed to detect α-methylacyl-CoA racemase (AMACR) in formalin-fixed, paraffin-embedded (FFPE) tissue sections.
The new tool is validated for use with a dual stain and VENTANA Basal Cell Cocktail (34ßE12+p63) to enable easy differentiation of morphologically difficult cases such as benign, atypical or cancerous on a single slide.
University of Utah develops robotic drill to minimise surgical time
Researchers at the University of Utah, US, developed an automated robotic drill with the potential to reduce complex cranial surgery time from two hours to two and a half minutes.
The computer-driven drill is able to make fast, clean and safe cuts, minimising the risk of infection, human error and surgical cost.
The device is claimed to make surgery 50x faster than standard procedures, which require the use of hand drills to make complicated openings.
Cardinal buys Medtronic’s Patient Recovery business for $6.1bn
Cardinal Health concluded the purchase of Patient Care, Deep Vein Thrombosis, and Nutritional Insufficiency business from Medtronic for a total of $6.1bn.
The transaction was funded with new senior unsecured notes of $4.5bn, existing cash, and borrowings under Cardinal’s current credit arrangements.
Initiated in April, the deal was subjected to regulatory approvals and other customary closing conditions.
Samsung launches new mobile full-body CT scanner
Samsung Electronics’ healthcare unit Samsung NeuroLogica launched a mobile, portable full-body computed tomography (CT) scanner, BodyTom Elite.
The CT system is an upgraded version of the firm’s BodyTom 32-slice CT scanner and features new visual design, upgraded software, hardware and workstation.
Hardware upgrades to the scanner include linear and step calibration, tilt sensor, step correction, quieter lift system and translate system, while the workstation improvements include battery backup, two-way patient communication system, better processing speed and export options.
New CUHK study shows early nasopharyngeal cancer detection with EBV DNA
A new study carried out by the Chinese University of Hong Kong (CUHK) indicated the possibility of using plasma Epstein-Barr virus (EBV) DNA analysis to detect early asymptomatic nasopharyngeal carcinoma (NPC).
Between 2013 and last year, researchers screened more than 20,000 middle-aged Chinese males without NPC symptoms in Hong Kong.
Upon plasma EBV DNA analysis, 309 subjects showed positive results during both initial and follow-up tests, before being further tested using nasal endoscopic examination and magnetic resonance imaging (MRI).
J&J opens new medical device centre in Texas, US
Johnson and Johnson (J&J) opened the new Centre for Device Innovation at Texas Medical Centre (CDI @ TMC) in the US to develop breakthrough medical devices.
The move comes as part of the collaboration between J&J Medical Devices Companies, J&J Innovation, and TMC.
To be directed by J&J Medical Devices Companies vice-president and cardiac surgeon William Cohn, the 25,500ft2 medical complex will feature a variety of amenities such as three-dimensional (3D) printers and scanners, laser cutters and a virtual reality room.
Boston Scientific agrees to buy Apama Medical for $300m
Boston Scientific signed an agreement worth up to $300m to acquire Apama Medical and its radiofrequency (RF) balloon catheter system designed for the treatment of a common heart rhythm disorder called atrial fibrillation (AF).
Affecting more than 33 million people across the world, AF is commonly treated with anti-arrhythmic drugs and cardiac ablation, which is a process where energy is delivered to the areas of the heart muscle causing an abnormal rhythm.
The agreement includes an initial upfront cash payment of $175m from Boston and an additional $125m in contingent payments between 2018 and 2020 based on clinical and regulatory milestones.





