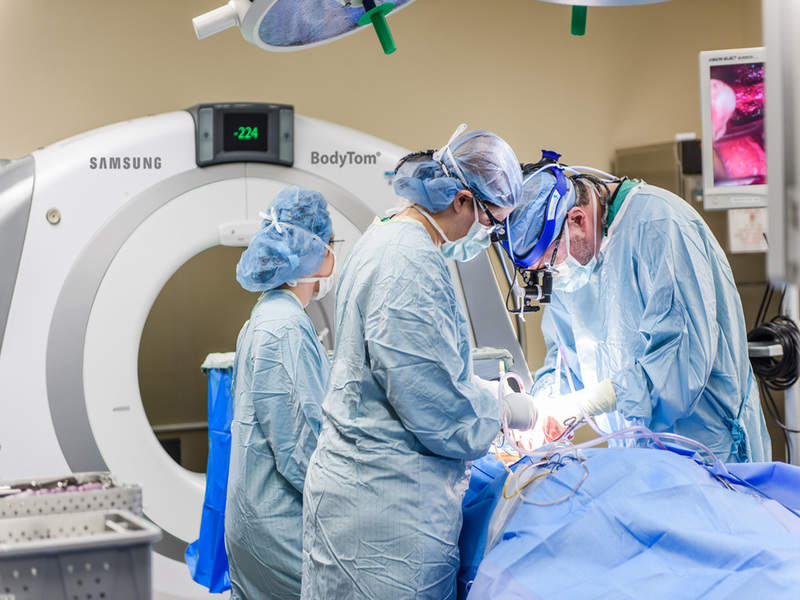

Samsung Electronics’ healthcare unit Samsung NeuroLogica has launched a new mobile, portable full-body computed tomography (CT) scanner, BodyTom Elite.
The new CT system is an upgraded version of the firm’s BodyTom 32-slice CT scanner and features new visual design, upgraded software, hardware and workstation.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Hardware upgrades to the scanner include linear and step calibration, tilt sensor, step correction, quieter lift system and translate system, while the workstation improvements include battery backup, two-way patient communication system, better processing speed and export options.
BodyTom Elite has received 510(k) clearance from the US Food and Drug Administration (FDA) last month.
Designed to accommodate patients of any size, the multi-departmental imaging solution comes with dose-structured reporting, CT dose check, AEC, paediatric, and adult reference protocols.
Samsung NeuroLogica CEO Philip Sullivan said: “We re-engineered the BodyTom Elite so that it more seamlessly integrates into any OR, neurosurgery, radiation oncology or radiology suite, and provides a more accurate, sharper image.”
In addition to wireless access point, the new system’s software includes low-dose lung cancer screening under 3mGy for the detection of abnormalities as well as contrast capability for use of CT angiography and perfusion imaging.
The metal artifact reduction (MAR) was added to improve metal implants and trauma cases, along with intuitive customisation, noise-reduction algorithm for improved image quality, 30 new custom kernels and automatic exposure control to adjust the amount of radiation according to patient characteristics.
Samsung NeuroLogica develops, manufactures and markets new imaging technologies and accurate diagnostic solutions for healthcare providers.
Image: Samsung BodyTom Elite. Photo: courtesy of Business Wire.



