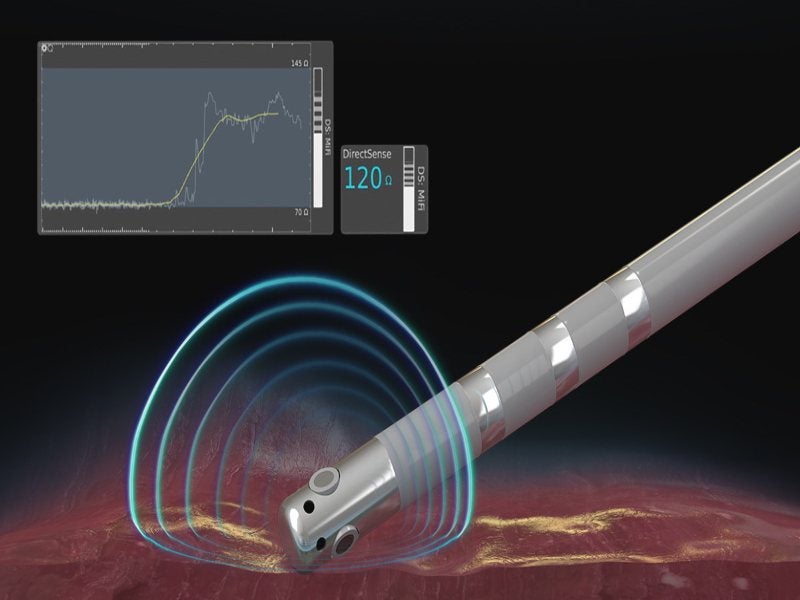
Boston Scientific has launched DIRECTSENSE Technology, a tool to monitor the effect of radio frequency (RF) energy delivery during cardiac ablation procedures.
Launched in the US, the tool received the US Food and Drug Administration (FDA) approval in April and is available on the RHYTHMIA HDx Mapping System.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It is considered to be the only tool to monitor changes in local impedance – electrical resistance – around the tip of the INTELLANAV MiFi Open-Irrigated (OI) ablation catheter.
Furthermore, the tool provides the clinician with an additional measurement of therapy effect during the ablation procedure.
Physicians use a catheter during the procedure to create lesions and destroy heart tissue that causes abnormal rhythms in patients with cardiac arrhythmias.
DIRECTSENSE Technology provides data on the impedance around the catheter tip to measure the ability of the tissue to respond to RF energy before physicians deliver therapy.
The tool also tracks the change in local impedance and provides physicians with a distinct understanding of tissue characteristics during an ablation. These insights may help to reduce the chances of over-ablation and avoid complications.
Boston Scientific Rhythm Management and Global Health Policy senior vice-president and chief medical officer Kenneth Stein said: “Building upon the success seen with the DIRECTSENSE Technology in Europe, we are pleased to introduce this tool to physicians and their patients in the US.
“This approval marks an exciting milestone for our growing electrophysiology portfolio, providing physicians with a more direct understanding of lesions and procedural efficiency to obtain optimal patient outcomes.”
The company recently received CE Mark for the POLARx Cryoablation System and has plans to launch the product in the coming months.
In December last year, it secured 510(k) clearance from the US FDA for its EXALT Model D duodenoscope.





