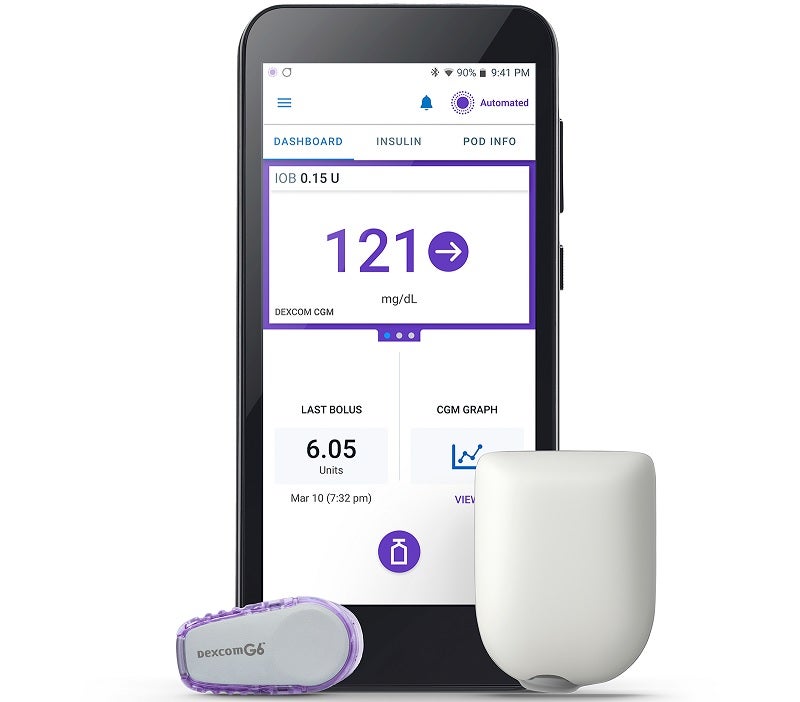
The US Food and Drug Administration (FDA) has approved Insulet’s Omnipod 5 Automated Insulin Delivery System (Omnipod 5) for type 1 diabetic patients aged six years and above.
The new system is claimed to be the first tubeless automated insulin delivery (AID) system.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It comes with a compatible smartphone and Dexcom G6 Continuous Glucose Monitor (CGM) System integration that helps protect patients from variations.
With SmartAdjust technology, Omnipod 5 includes a tubeless insulin Pod, Dexcom G6 CGM and the Omnipod 5 mobile app, along with an integrated SmartBolus Calculator.
The device is designed to manage glucose without fingersticks, tubes or multiple daily injections.
Insulet president and CEO Shacey Petrovic said: “Omnipod 5 is a life-changing technology that we believe will revolutionise the market and the lives of people with diabetes.
“We are incredibly proud of this simple-to-use, elegant system, designed to deliver unmatched freedom and to greatly simplify insulin management and improve glucose control for our users.”
Users can download the mobile app on a personal smartphone or use the Omnipod 5 Controller, which is provided free with the first prescription.
The company stated that SmartAdjust receives a Dexcom CGM value and trend every five minutes and predicts where the users’ glucose level will be in the next 60 minutes.
Using this information, the Omnipod 5 system increases, decreases or pauses the delivery of insulin using the patient’s desired and customised glucose target.
This helps protect patients against highs and lows.
Insulet will launch the Omnipod 5 system through the pharmacy channel and later offer it in a limited market release.
Shortly after the limited market release, the system is expected to be broadly available in the market.
Last March, the company reported positive data from the first pivotal trial of the Omnipod 5 system.




