PeekMed has received 510(k) clearance from the US Food and Drug Administration (FDA) for its new automated orthopaedic solution.
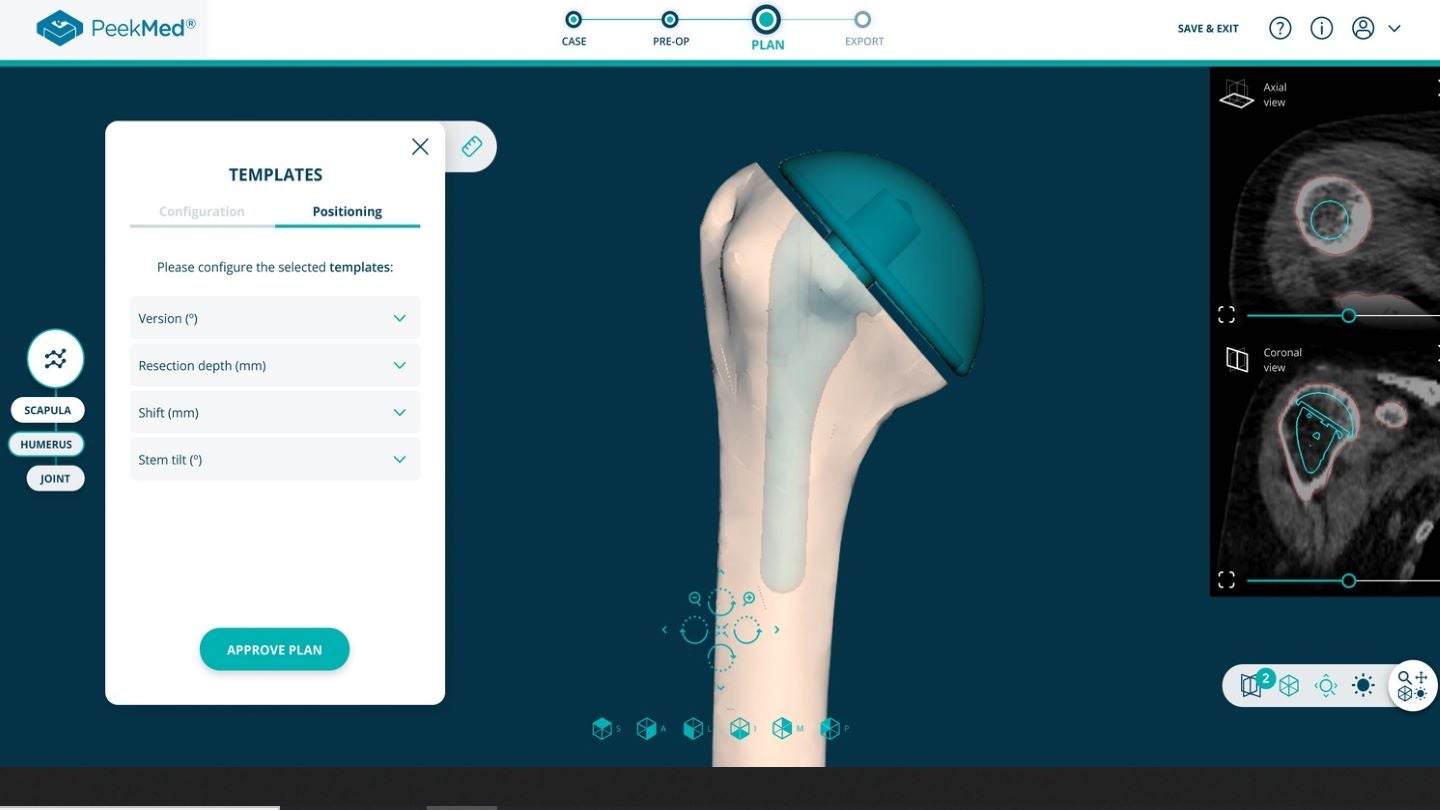
Using artificial intelligence (AI) and deep learning technologies, the new web-based solution enables live automatic segmentation of computerised tomography (CT) scans and X-Rays. It also provides automatic planning and landmark detection.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
With the support of these technologies, the new solution can provide a live plan for surgeons within 30 seconds.
According to the company, all the planning steps are automatic and only need approval from the surgeon, who can also carry out changes if required.
By representing medical images in a 2D or 3D environment, PeekMed web can carry out a total overview of the surgery.
The solution can also perform relevant measurements on images and add templates.
Furthermore, the new web-based automated planning solution integrates all previous PeekMed features and enables easy connection to other orthopaedic solutions in common anatomical regions of the adult musculoskeletal system.
These regions include the knees, hips and upper limbs.
PeekMed chairman and CEO João Pedro Ribeiro said: “With this FDA clearance, PeekMed is ready, once again, to be a game-changer in the integration of technology with orthopaedics.
“Having the ability to get a plan in less than 30 seconds is just incredible and a huge achievement for our team.
“We understood the pains of our surgeons, who were waiting hours, or sometimes days, to get a case planned. We aim to solve those challenges!”
The company plans to showcase the new automated orthopaedic solution at the American Academy of Orthopaedic Surgeons Annual Meeting (AAOS), which is being held between 7 and 11 March in Las Vegas, Nevada, US.





