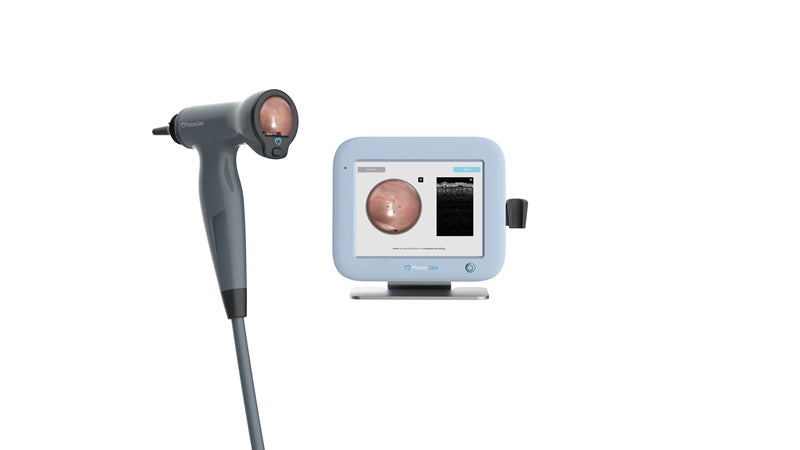
The US Food and Drug Administration (FDA) has granted 510(k) clearance for PhotoniCare’s TOMi Scope for non-invasive imaging of the middle ear to diagnose ear infections.
TOMi Scope is a hand-held, non-invasive imaging otoscope that allows clinicians to check for the presence or absence of fluids in the middle ear, one of the principal indications of childhood ear infections.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
By leveraging optical coherence tomography (OCT) high-resolution depth imaging, TOMi Scope can determine whether the fluid is present in the middle ear and characterise its type and density, even in cases with substantial wax build-up.
Furthermore, the OCT scanner provides the physicians with a real-time view of middle ear fluid, as well as high-resolution video images of the eardrum surface.
PhotoniCare co-founder and CEO Ryan Shelton said: “At PhotoniCare we set out to solve the massive problem of frequent misdiagnosis of middle ear infections and the overuse of antibiotics and referrals to surgery in children that result.
“We thank the FDA for clearing our TOMi Scope under a new product code unique to our technology, and look forward to bringing this innovation to doctors and patients very soon.”
Middle ear infections affect more than 80% of children, often misdiagnosed up to half of the time following examination with a conventional otoscope.
Misdiagnoses result in recurrent infections, as well as the use of increasingly potent antibiotics and surgery.
Each year, more than one million children in the US undergo surgeries to drain fluid from the middle ear.
PhotoniCare said it plans to begin its launch of the TOMi Scope at selective locations in the US soon with a national rollout planned for later in the year.



