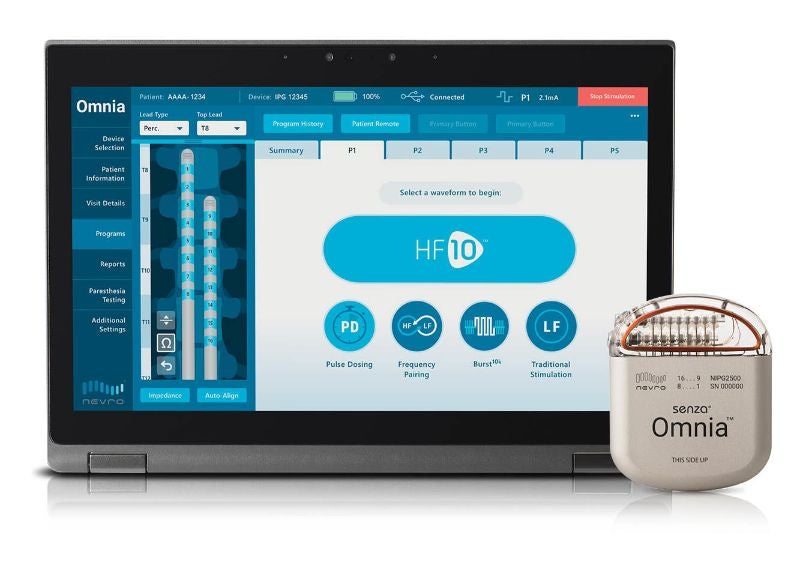
Medical device company Nevro has launched Senza Omnia Spinal Cord Stimulation (SCS) System to treat chronic pain in the US, following the US Food and Drug Administration (FDA) approval.
The system delivers SCS frequencies between 2Hz and 10,000Hz, along with the company’s HF10 therapy, which combines a 10kHz frequency and a treatment algorithm.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
HF10 is a non-pharmacologic neuromodulation platform to treat chronic pain.
Senza Omnia consists of a programmer to allow physicians to provide a variety of waveforms, separately or in combination, across the SCS frequency spectrum.
The device comes with a patient remote to support extended programming capacity. It enables expansion on the HF10 algorithm, where physicians can offer additional waveforms.
Nevro integrated Senza Omnia with an upgradeable implantable pulse generator, which can last for more than ten years, irrespective of the stimulation profile.
The company added that the system has conditional full-body MRI approval, with the potential to upgrade to future waveforms and frequencies.
Nevro chairman and CEO Keith Grossman said: “The Omnia system was developed based on feedback from the pain community and has resulted in a product that Nevro is uniquely able to provide.
“Omnia not only offers HF10, the most studied therapy available in the market that physicians have provided to over 53,000 patients but it also gives physicians access to all other SCS frequencies, either independently or paired with HF10.”
The company expects to secure regulatory approvals in Europe, as well as Australia next year.
Nevro initially developed the Senza SCS system for non-pharmacologic treatment of chronic pain. Its portfolio currently includes the Senza System, Senza II System and Senza Omnia System.



