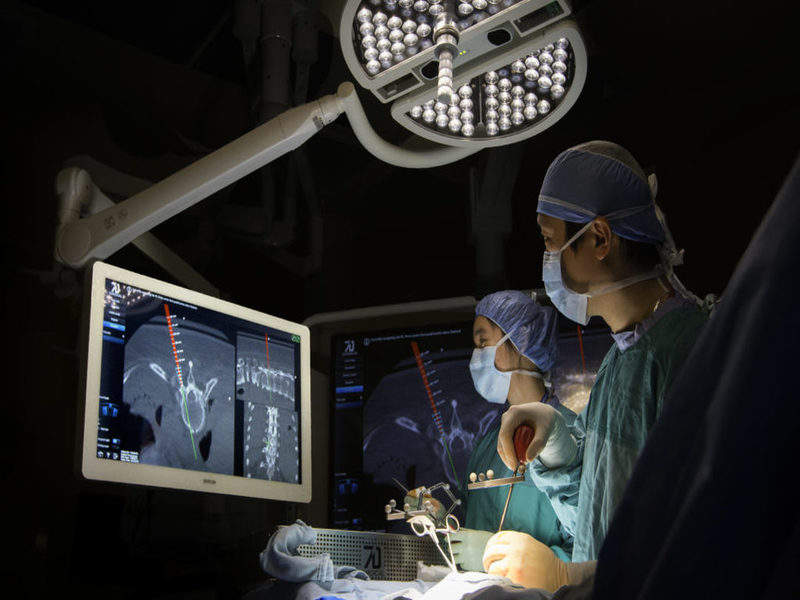

7D Surgical has received the US Food and Drug Administration (FDA) 510(k) clearance and Health Canada medical device license (MDL) for the use of its Machine-vision Image Guided Surgery (MIGS) system in spine surgery.
The system is equipped with 3D optical technologies and machine vision algorithms allowing easy registration of spinal surgery patients automatically with visible light, contrary to intraoperative radiation systems.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The firm claims that the Flash Registration of the system enables de novo spinal registration in less than 20 seconds, eliminating the possible 30 minute interupption of surgery due to the registration process.
7D Surgical chief executive officer Beau Standish said: “When navigating the spine, surgeons traditionally have had two time-consuming and expensive IGS options: systems that rely on intraoperative radiation emitting devices or systems that utilise laborious manual point matching techniques.
“We believe the inefficiencies of these systems have limited the adoption of IGS in spine procedures to less than 20%.
“7D Surgical’s MIGS system has now removed these barriers, providing surgeons and their hospitals with a superior product option.”
The prototype of MIGS technology has been evaluated in 160 patients in clinical trials at Sunnybrook Health Sciences Centre in Toronto, Canada.
The MIGS navigation technology is fixed to an onboard overhead surgical light to provide better line of sight. It employs simplified software, which can be controlled by the surgeon with a foot pedal.
The clearance from both the US and Canada enables the launch of MIGS throughout North America.
Image: Surgeons looking at MIGS images. Photo: courtesy of 7D Surgical.





