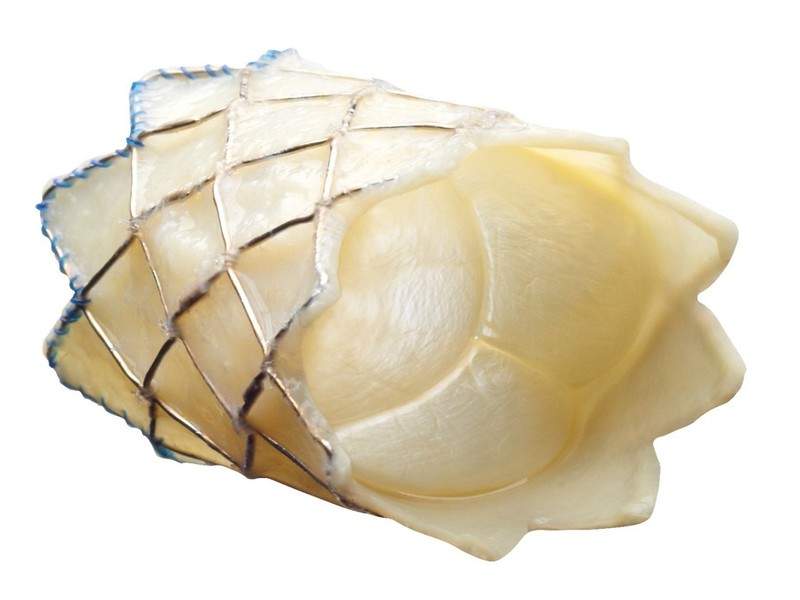

Irish medical technology firm Medtronic has obtained the US Food and Drug Administration (FDA) approval for its Melody transcatheter pulmonary valve (TPV) for use in patients whose surgical bioprosthetic pulmonary heart valves have failed.
The new TPV is designed for the pulmonic position to prolong the time between open-heart surgeries for patients with a dysfunctional right ventricular outflow tract (RVOT) conduit caused by congenital heart disease (CHD).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Intended to be a minimally invasive treatment option, the valve is placed inside a failing pulmonic surgical heart valve through a low-profile, delivery catheter called Ensemble II delivery system that is specifically designed to deliver the TPV.
Medtronic Heart Valve Therapies business vice-president and general manager Rhonda Robb said: "Unlike other transcatheter valves currently on the market, Melody TPV is uniquely designed for use in the pulmonic position and is thus well suited for implantation in failed surgical pulmonary heart valves.
"This approval further demonstrates our commitment to improving treatment options for congenital heart disease and we look forward to bringing this proven non-surgical option to congenital patients."
While the transcatheter has received CE mark in September 2006, it was launched in the US in 2010, and has been implanted in more than 10,500 patients till date.
Seperately, the company has also secured expanded 510(k) clearance from the FDA for its Kyphon Xpede bone cement to fix pathological fractures of the sacral vertebral body or ala through sacral vertebroplasty or sacroplasty.
The bone cement was previously indicated for treatment of vertebral fractures associated with osteoporosis, cancer or benign lesions.
Image: Melody transcatheter pulmonary valve. Photo: courtesy of Medtronic.





