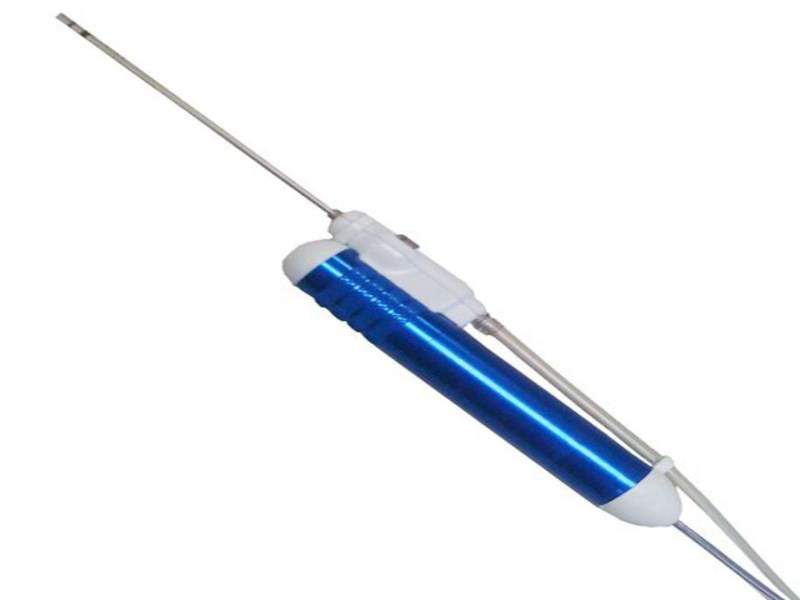

NICO has received a new and updated 510(k) clearance from the US Food and Drug Administration (FDA) for two of its patented brain surgery technologies Myriad and BrainPath.
Myriad it is an automated, multi-functional tool used for precise resection, suction, clot evacuation and tumour removal in neurosurgery, spinal surgery, ENT and otolaryngology.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Approximately 12,000 procedures have been already performed using this technology.
The FDA has updated the recently received clearance for Myriad automated tissue removal technology use in specific neurosurgical indications, to use in primary and secondary brain tumors, vascular abnormalities and malformations, and intraventricular tumors and cysts.
NICO president and chief executive officer Jim Pearson said: “NICO has spent the last five years bringing together key technology elements to enable minimally invasive neurosurgery for subcortical tumours and clots.
“The goal of NICO has always been to create technology that could create less deficits and faster recoveries in brain surgery.
“The clinical evidence shows this is possible, and we believe that is directly related to finding less disruptive ways to access the brain that were not possible before.
“And we believe it will revolutionise the intracranial neurosurgical market.”
With initial clearance secured in July 2015, BrainPath access technology is now cleared for use in specific disease conditions such as high grade gliomas and Glioblastoma Multiforme, secondary metastatic tumours, as well as vascular abnormalities like intracerebral hemorrhage.
Myriad and BrainPath can be combined for non-invasive access, automated tumour removal and clot evacuation near critical structures along with collection and preservation of removed tissue and clots.
Image: NICO Myriad. Photo: courtesy of NICO Corporation.





