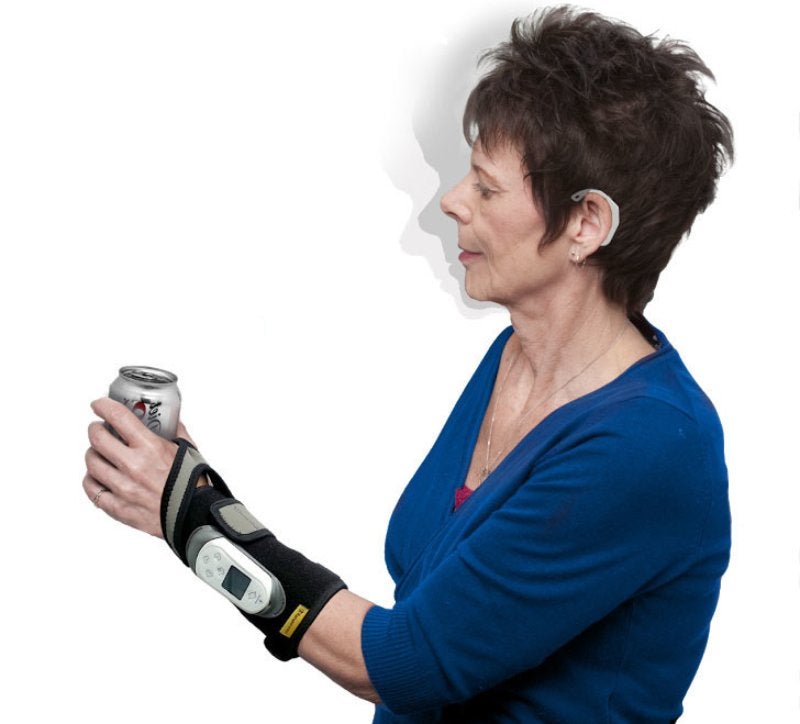
Rehabtronics has announced that its ReGrasp Bionic Glove, a rehabilitation device to improve hand mobility, is now available in the US.
The US Food and Drug Administration (FDA)-cleared ReGrasp leverages functional electrical stimulation (FES) technology to enhance hand function after stroke or injury. It can be used by patients at home or in a clinical setting.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Rehabtronics CEO Dr Rahul Samant said: “Until now, rehabilitation gloves with FES technology have only been accessible to patients in a clinical setting.
“Our research indicates that patients regain hand use faster when using ReGrasp for continuous therapy at home and functional use everywhere. That’s why we’re making ReGrasp available directly to patients through our website.”
The device can be purchased with a prescription from a physician or qualified therapist.
ReGrasp comes with a discrete wireless earpiece that allows patients wearing the bionic glove to control their hand function using simple head movements.
Additionally, the device’s FES unit is programmable, enabling the addition of home exercise programmes. This will help patients to use the glove to carry out daily tasks.
Home health therapist Ashley Gatewood said: “The ReGrasp provides patients with the opportunity to have functional rehabilitation at their fingertips in order to improve their independence and quality of life.
“This device opens the door for patients to have access to advanced technology in their home, rather than just in a clinical setting.”
Established in 2003, Rehabtronics focuses on developing rehabilitation devices for people who are paralysed or immobile. The company’s latest product is Prelivia, which is designed to address pressure injuries.





