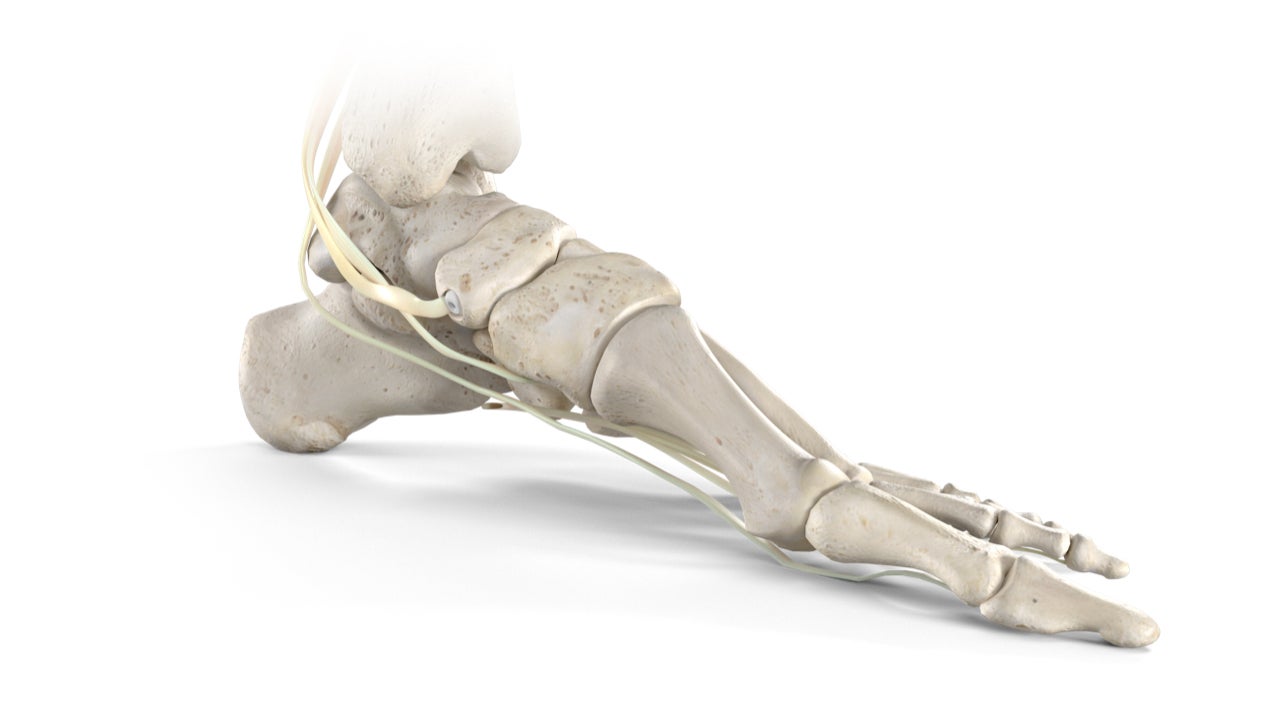
Medical technology company Stryker has introduced the new Citrelock Tendon Fixation Device System, which is designed to enable fixation without causing damage to the tendon during placement.
The new system consists of a tendon thread with a resorbable technology called Citregen.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Citregen is made up of citrate, calcium and phosphate molecules that are essential to the bone anatomy and offers differentiated chemical and mechanical properties for use in orthopaedic surgeries.
With a controlled and homogeneous resorption process, the Citrelock system is intended to prevent bulk degradation and chronic inflammation.
The system also provides surgeons with strength that is similar to cortical bone and a modulus in line with cancellous bone.
In addition, the Citrelock system comes with a material polymer structure that is comparable to the extracellular matrix protein network.
During the healing phase, structural integrity is maintained by Citregen and the host tissue replaces the implant over time.
Stryker foot and ankle business marketing and medical education vice-president Michael Rankin said: “Our customers are looking for a more predictable and effective bioresorbable material than what is currently on the market.
“The Citrelock Tendon Fixation Device System helps fill that need with its innovative material technology and unique design.
“Citregen is an exciting addition to Stryker’s existing biomaterial portfolio and will be expanded for use in additional Trauma and Extremities indications in the future.”
Stryker will launch the Citrelock system at the American Orthopaedic Foot and Ankle Society (AOFAS) annual meeting, to be held from 22-25 September 2021 in Charlotte, North Carolina, US.
Along with Citrelock, the company is planning a new product reception for other recent additions to its Foot and Ankle portfolio.
Stryker Foot and Ankle portfolio includes plating platforms, screw systems, hammertoe solutions, staple solutions, external fixation systems and titanium implants.
In July, the company received US Food and Drug Administration clearance for its InSpace balloon implant for arthroscopic therapy of massive irreparable rotator cuff tears.



