Therma Bright has created a new venture called Torion Biosciences to develop, license, and commercialise a rapid antigen test, CoviSafe, for screening individuals infected or suspected of exposure to Covid-19 virus.
The company will have 50% control over Torion while the remaining 50% will be owned by others, including veteran biotech executives as the primary shareholders of the new venture.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Furthermore, Therma Bright ended its co-development deal with Orpheus Medica and Torion will hold discussions with key personnel from Orpheus, depending on the needs.
Currently, Torion is in late-stage discussions to buy a series of biological candidates as primary reagents for CoviSafe.
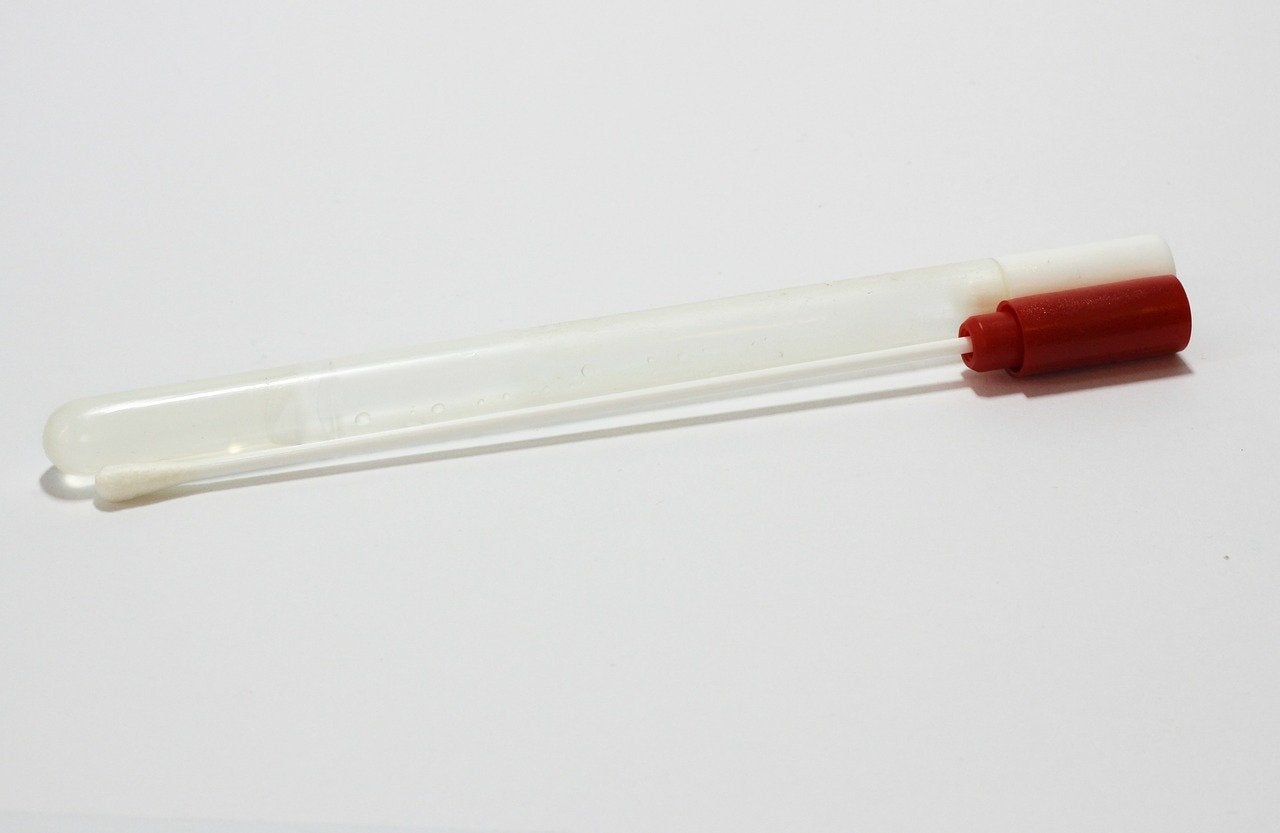
Developed as a point-of-care cassette, CoviSafe is a rapid antigen test that does not require lab instruments or specialised lab personnel to be carried out.
The device uses lateral flow technology to detect the presence of the Covid-19 virus in saliva or other swab samples, including upper respiratory samples, and can provide results in 15 minutes.
CoviSafe is developed for use at point-of-care settings such as long-term care facilities, schools, or even at home. It will be progressed into patient sample validation, and later, on to clinical studies with the newly acquired biological molecules.
nanoComposix will carry out CoviSafe product development.
Therma Bright will seek Food and Drug Administration (FDA), Health Canada, and the CE mark for fast track or expedited approval for CoviSafe to test symptomatic patients.
Therma Bright CEO Rob Fia said: “By creating Torion, Therma Bright will own a significant percentage of the venture and the future commercial sales of CoviSafe.
“Our capacity, through our partners, will enable us to manufacture pilot test kits for feasibility and research purposes very quickly.”





