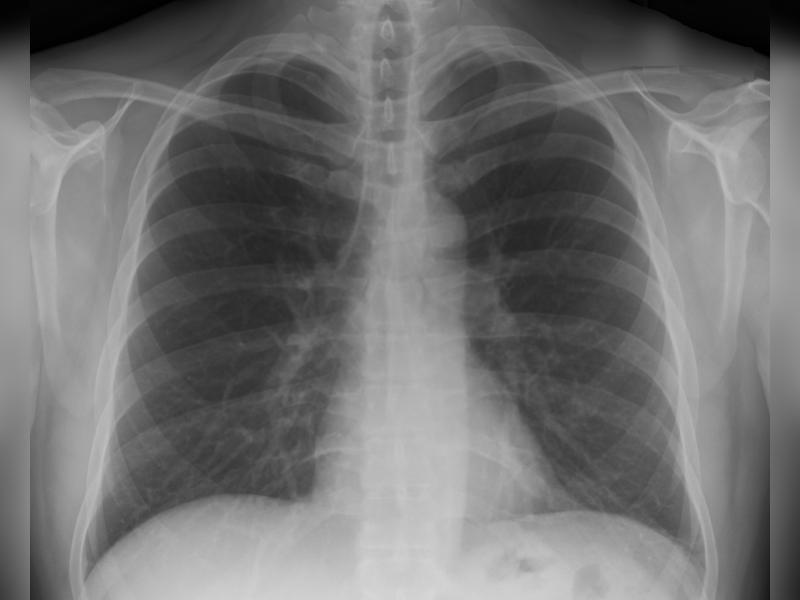
The US Food and Drug Administration (FDA) has granted regulatory clearance to 4DMedical to market its four-dimensional lung imaging process XV Technology.
The technology is a software-as-a-service (SaaS) diagnostic tool available through secure cloud subscription. It can automatically analyse any functional lung impairment from a single X-ray.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
4DMedical founder and CEO Andreas Fouras said: “Our XV Technology is a valuable new respiratory diagnostic tool.
“It provides critical information about the functional and structural state of a patient’s lungs in the treatment of illnesses such as Covid-19, asthma, chronic obstructive pulmonary disease, cystic fibrosis and lung cancer.
“FDA clearance means we can fast-track our ‘go-to-market’ strategy to ensure hospitals and doctors in the United States have almost immediate access to our XV Technology Ventilation Reports.”
XV Technology can be deployed immediately, using the existing hospital and clinical infrastructure with no capital expenditure or training required, the company noted.
The process requires imaging departments to electronically send an X-ray, which is taken using existing fluoroscopy equipment, to 4DMedical.
4DMedical software identifies and quantifies any functional impairment by rapidly and automatically analysing and applying its proprietary algorithms to the X-ray.
Within three hours, the software provides a ventilation report to the hospital, enabling clinicians to choose the most effective treatment and allocate hospital resources.
The company noted that its ventilation reports can deliver quantitative support for Covid-19 diagnosis and follow up examinations for patients with or recovering from Covid-19.
However, XV Technology is not intended to replace molecular tests as the primary diagnosis method for Covid-19.
XV Technology supported by 15 years of preclinical and clinical trials and existing collaborations with hospitals across the world.
Australian government’s Medical Research Future Fund (MRFF) Frontier Health and Medical Research Programme has validated the technology through an extensive vetting process.





