Netherlands-based medical device company Wright Medical Group has signed an agreement to acquire 100% outstanding shares of US-based orthopaedic devices firm Cartiva in a $435m all-cash deal.
Cartiva primarily develops medical devices to treat osteoarthritis.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
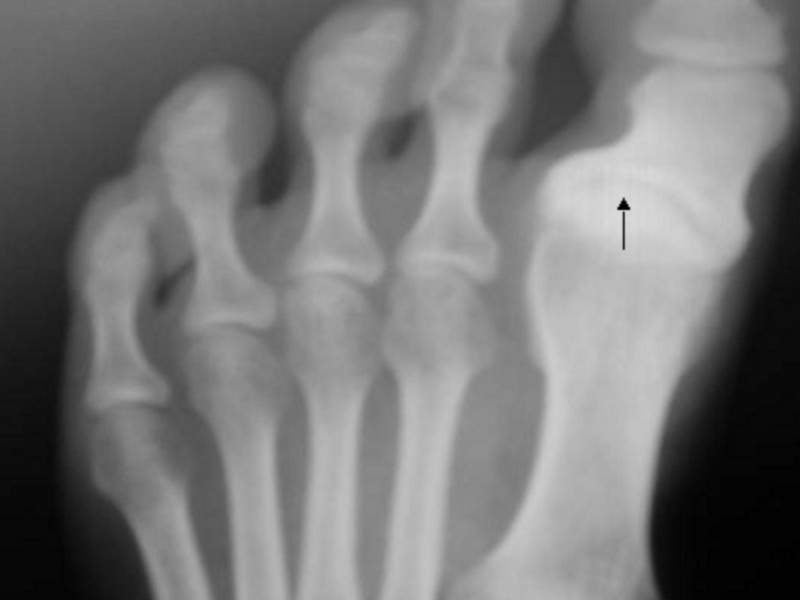
The acquisition will expand Wright Medical’s global Extremities unit with the addition of Cartiva’s technology, Synthetic Cartilage Implant (SCI), which is designed for a high-volume foot and ankle procedure.
In July 2016, the SCI secured US premarket approval (PMA) for the treatment of arthritis at the base of the great toe. It also has gained approvals in Canada, EU, Brazil, Chile and Australia.
The product contains an organic polymer that is biocompatible and durable with low friction.
The polymer’s functionality is similar to that of the natural cartilage and allows implantation in approximately 35min.
Cartiva is claimed to mitigate joint pain while retaining the foot’s natural movement, along with mobility and range of motion. This enables patients to return to their daily functions and activities faster when compared to those treated through a fusion procedure.
Wright Medical expects the deal to bolster growth opportunities for the Extremities business.
Wright Medical Group president and CEO Robert Palmisano said: “Supported by compelling clinical performance and the only product of its kind backed by Level I clinical evidence, Cartiva is experiencing rapid commercial adoption and is well-positioned for future growth as it addresses large markets with significant unmet needs.
“We believe this technology is a perfect fit for our Lower Extremities business and adds a differentiated product that addresses a common condition that is treated by most foot and ankle surgeons and has strong patient demand.”
Subject to customary closing conditions, the acquisition is scheduled to complete in the fourth quarter of this year. Following the closure of the deal, Wright expects the transaction to increase its net sales in 2019 by $47m.





