Delphinus’ SoftVue™ is a fast, fully-automated 3D whole breast ultrasound tomography system first-ever developed in medical imaging for dense breast cancer screening in addition to mammography.
The SoftVue™ system represents a breakthrough in tissue characterisation based on transmission signals and satisfies the unmet clinical requirements for early breast cancer detection in asymptomatic women with dense breast tissue. It also provides a unique annual screening solution for the group.
SoftVue™ received premarket approval (PMA) from the US Food and Drug Administration (FDA) in October 2021.
SoftVue 3D breast ultrasound tomography system features and technology
The SoftVue™ system is designed to be used as a B-mode ultrasonic imaging device and is not an alternative to mammography.
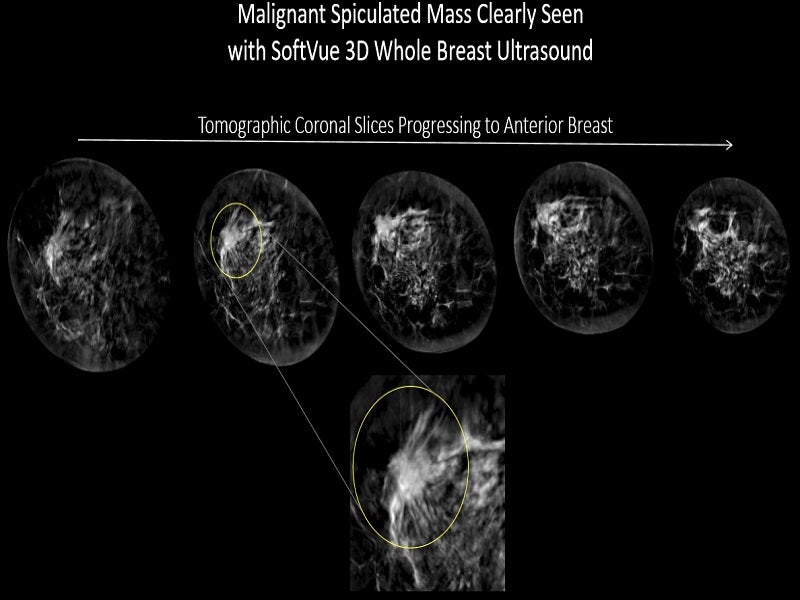
The system features a circular transducer of 22cm diameter and proprietary algorithms that drive its TriAD™ triple acoustic detection technology. The transducer is configured with more than 2,000 transducer elements arranged in a unique uniform circular ring.
Each transducer element transmits and receives ultrasound signals around the breast in a sequenced circular array, providing a complete 360° coverage.
TriAD™ technology records, speed and direction of sound waves, and attenuation. The reflection echoes outline the anatomic detail while the transmission signals passing through the breast tissue quantitatively determine the sound speed and the signals interacting with the breast tissue provide attenuation.
The technology offers unrivalled images of the breast tissue structure and provides tissue attributes of sound speed and attenuation to help physicians in distinguishing healthy tissue from the concerned regions.
Sequr™ Breast Interface details
The Sequr™ Breast Interface is an anatomically formed guidance system that is an essential component of the SoftVue™ system. It is a gentle, disposable gel pad that engages with the front of the breast to align, steady and centre it during the imaging process, supporting breast contour adjustment and proper positioning of the breast tissue for efficient image acquisition.
The system incorporates a padded tabletop, custom cushions and soft membranes to promote a gentle experience for patients.
SoftVue™ test procedure
During the breast examination, the patient lies on the stomach on the padded table, submerging the breast to be examined in a warm water bath. The breast is gently stabilised and supported by the Sequr™ Breast Interface gel pad.
The 360º ring transducer encircles and scans the breast from chest wall to nipple in three minutes and captures new images in 2mm intervals.
The system’s incredible imaging capability not only records reflected echoes but also measures signals transmitted through the breast.
The recorded signals are subsequently analysed by sophisticated algorithms, offering cross-sectional slices of the complete volume of breast tissue.
SoftVue 3D breast ultrasound tomography system benefits
SoftVue™ is a new approach for imaging the entire breast utilising sound and water, which make the solution safe and gentle. It improves dense breast screening and detects up to 20% more breast cancers with higher accuracy and potentially lesser biopsies than full-field digital mammography (FFDM) alone, suggests clinical evidence.
The examinations are performed at the same visit as mammograms screening, promoting more efficient workflow and faster delivery of results.
The entire scanning process takes between two and four minutes for each breast without compressing the breast or exposing it to radiation. The scan is private and discrete, and the breast is not visible throughout the scanning procedure.
Prospective case collection (PCC) study
Delphinus conducted a prospective case collection (PCC) study using the SoftVue™ system to establish an image database for both cancer and non-cancer cases. The cases were subsequently utilised in a series of multi-reader, multi-case (MRMC) experiments conducted by the University of Chicago.
The PCC programme started in 2017 and enrolled more than 8,500 asymptomatic individuals in Breast Imaging Reporting and Database System (BI-RADS) c or d density groups at ten prominent breast imaging facilities across the US.
More than three million coronal plane images generated by the project were analysed by a team of radiologists.
The pivotal MRMC showed that SoftVue™ identifies and differentiates between normal (BI-RADS 1 and 2) and abnormal (BI-RADS 3 and 4) lesions with 20% more sensitivity and 8% more specificity over FFDM alone.