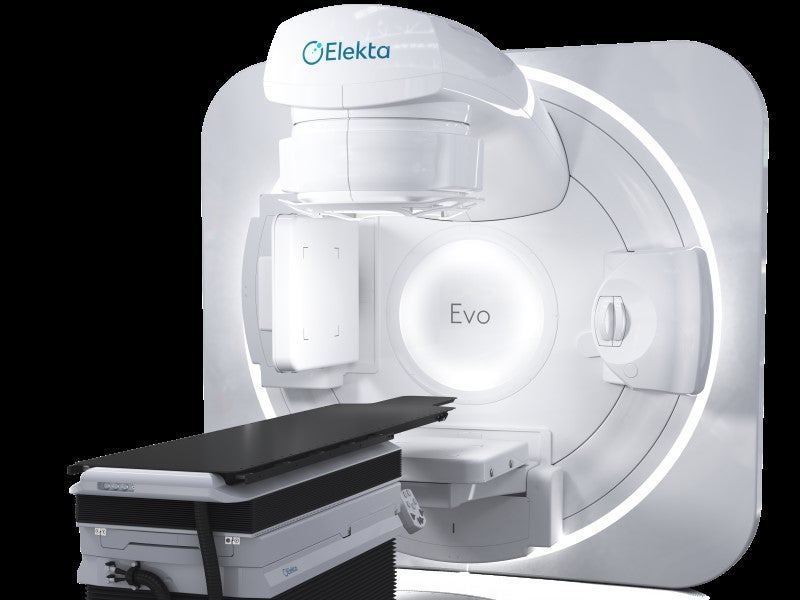
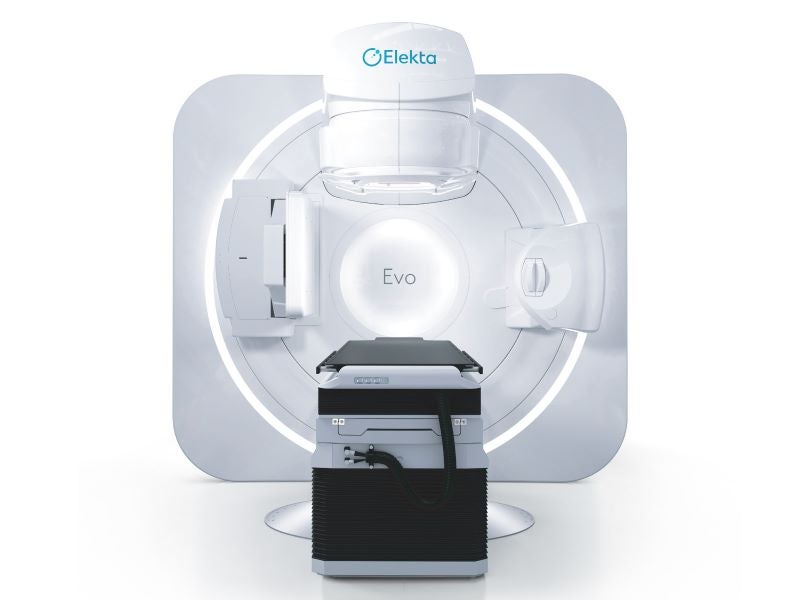
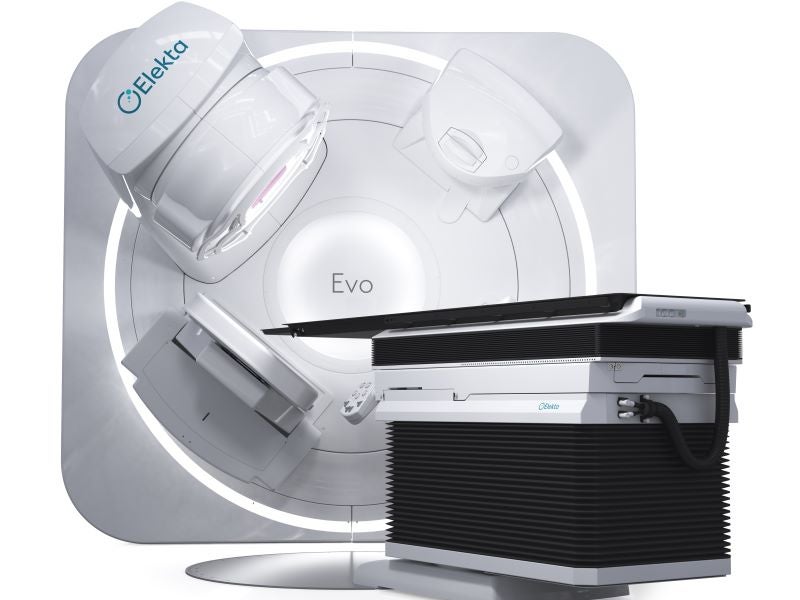
Elekta Evo is a next‑generation, AI-enabled, computed tomography-guided linear accelerator (CT‑Linac) developed by Elekta, a precision radiation therapy equipment company based in Sweden.
The device is intended to support the wider integration of adaptive radiotherapy into everyday clinical oncology-specific workflows.
Evo was unveiled at Europe’s largest radiation oncology industry exhibition, the Annual Congress of the European Society for Radiotherapy and Oncology, held in Glasgow, Scotland, in May 2024.
Regulatory approvals for Elekta Evo
Elekta Evo received its Conformité Européenne (CE) mark in Europe in September 2024.
In December 2025, Evo obtained registration from India’s Central Drugs Standard Control Organisation, with initially limited international availability.
The device subsequently secured 510(k) clearance from the US Food and Drug Administration in January 2026, enabling its use by radiation oncology professionals in the US.
Elekta Evo design and features
Elekta Evo integrates a comprehensive range of ultra‑precision radiotherapy techniques within a single platform. It features advanced stereotactic capabilities including flattening filter‑free (FFF) beams, 6D couch positioning and 1mm resolution for detailed dose sculpting and optimised stereotactic radiosurgery (SRS).
The system operates in two distinct lighting modes, Moonlight mode and Treatment mode. Moonlight mode is configured to support quality assurance activities and patient set‑up, while Treatment mode is configured with a focus on patient comfort during delivery.
A maximum leaf speed of up to 6.5cm/s, in combination with a dynamic Y jaw speed of 9cm/s and high dose rate operation (6 FFF at 1,400 monitor units (MU)/min and 10 FFF at 2200MU/min), supports SRS and stereotactic body radiotherapy (SBRT) within standard treatment time slots.
The system is designed to enhance organs‑at‑risk sparing through high‑definition beam shaping and very low multileaf collimator leaf transmission, helping to limit radiation dose to surrounding healthy tissue.
Using a 40cm × 40cm field and IntelliBeam’s variable control point technology, Elekta Evo enables Volumetric Modulated Arc Therapy (VMAT) delivery in treatment times reported to be up to 70% shorter than with conventional techniques.
The system is configured with two key components: the Iris imaging system, which is designed to improve visualisation of target structures, and Elekta ONE® Online software, which supports distributed treatment planning.
Evo Iris imaging system
The Evo Iris’ high‑definition, AI‑enhanced imaging is driven by AI-led scatter correction alongside advanced reconstruction algorithms.
The high‑quality images produced by the system support the visualisation of tumours and organs at risk, facilitating precise contouring. Iris is designed to provide a clearer depiction of target volumes and critical structures for each treatment fraction.
The system’s synthetic‑free imaging approach is intended to improve Hounsfield unit accuracy, supporting more precise treatment planning and adaptation.
Elekta ONE software suite
The Elekta ONE® Online consists of multiple Elekta software solutions integrated with the CT‑Linac. It provides distributed treatment planning, accelerated dose calculation, and AI‑supported automation for contouring and dose planning.
To further support optimisation of both offline and online treatment planning, Elekta has added an extension to this portfolio: Elekta ONE Planning, powered by MIM.
Elekta ONE Planning offers distributed planning, faster dose computation, and AI‑assisted tools for contouring and treatment plan generation. The platform is designed to be compatible with both Elekta and non‑Elekta systems including proton therapy devices.
Benefits of Elekta Evo
The Elekta Evo device supports the full range of external beam radiotherapy (EBRT) delivery techniques including SRS, SBRT, intensity modulated radiation therapy, VMAT, multiple photon energies and electrons, with precision integrated into the system design.
The system provides both offline and online treatment plan adaptation, together with enhanced image‑guided radiation therapy (IGRT) capabilities. The EBRT and IGRT options allow clinicians to use a comprehensive set of treatment approaches tailored to the specific requirements of each cancer patient’s case or individual treatment session.