A unique identifier applied directly onto an instrument, and available at any time to facilitate its tracking and computerised management, is a real advantage for health institutions.
Controlling the traceability of invasive medical appliances is a significant challenge facing health institutions. Sterilisation departments have put in place identification systems ranging from double labels, transferable to the patient’s case file, to computerised systems using bar-codes read by electronic scanners.
For surgical instruments, most institutions use labels attached to their surgical bags or on the instrument itself. These two methods add:
- Additional handling in the operative phase (opening the bags or removing the label from the instrument)
- Reproduction of the identification process, using up a consumable, at each medical procedure
The problem for Technifor, therefore, was to find a reliable method of feeding down traceability to instrument level, and to provide a comprehensive record of the successive stages in the life of each instrument until it is discarded.
Direct part marking identification system
The challenge: propose a direct part marking identification system, enabling information to be captured automatically with no risk of input error, create a link between the information thus captured and the computerised management system for the complete instrument inventory.
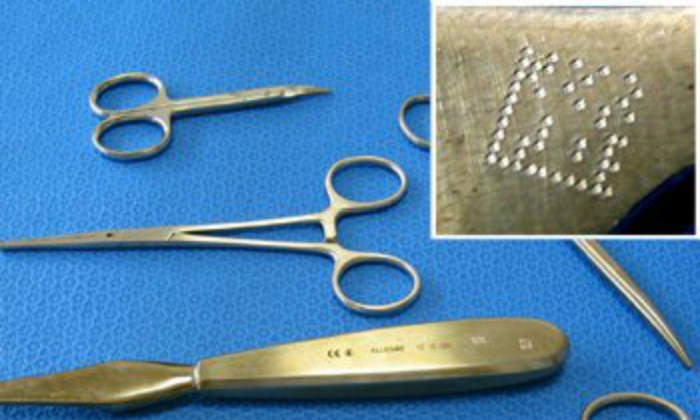
Technifor was able to demonstrate the quality of a solution whereby the instrument would be identified by a two-dimensional code, readable by Vision tools: DataMatrix. This compact 2D code enables up to 20 characters to be stored within a tiny area 3mm square.
Micro-percussion marking
With reference to marking, at its test centre and in cooperation with French Hospitalisation Centre, Technifor applied its skills in a variety of technologies and confirmed the possibility of using direct marking by micro-percussion. Marking by micro-percussion presents excellent resistance to the rigorous sterilisation process.
Set up in a stand-alone marking station with two hand-held scanners, this solution is now running in several hospitals, and is providing total satisfaction in terms of the internal traceability and management of the hospital’s medical appliances.