Alleima, headquartered in Sandviken, Sweden is listed on Nasdaq Stockholm as of August 31, 2022. The company’s origins date back to 1862. The separation from Sandvik has made it possible to operate as an independent entity enabling Alleima to execute its strategy of driving growth, materials innovation, operational and commercial excellence, and sustainability.
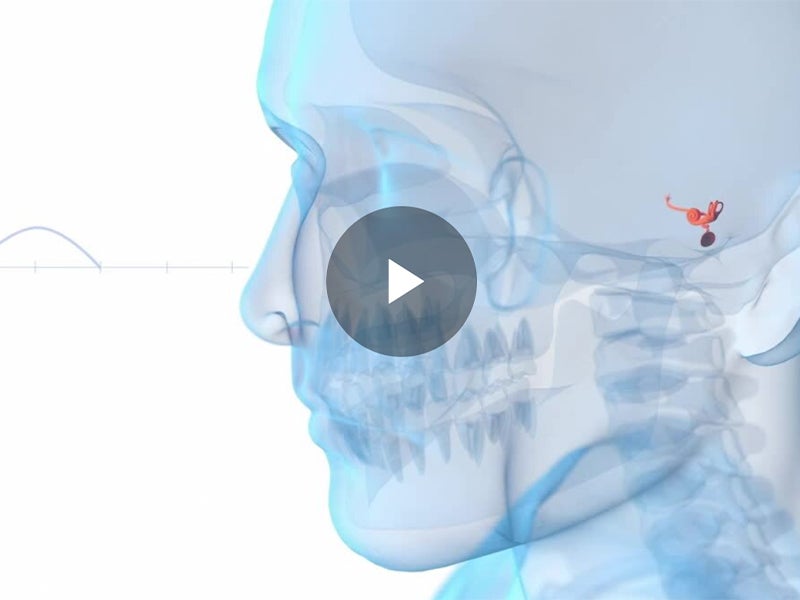
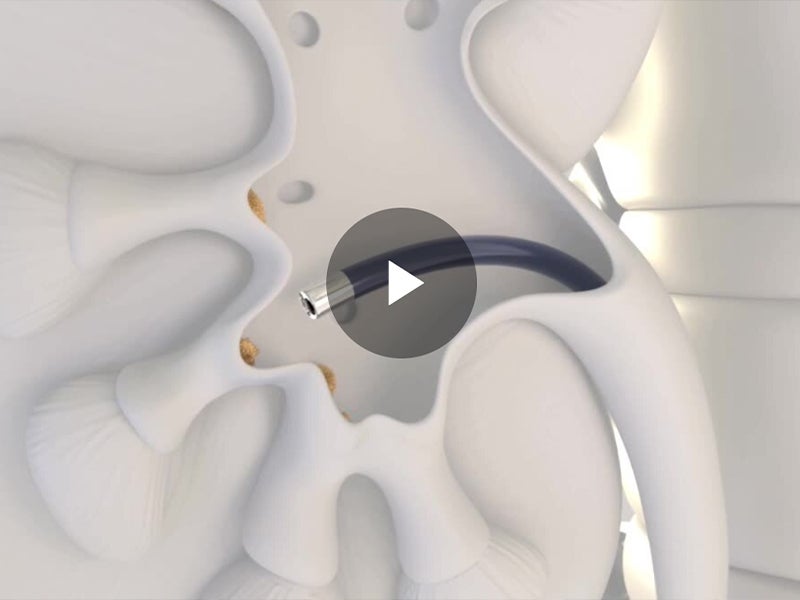
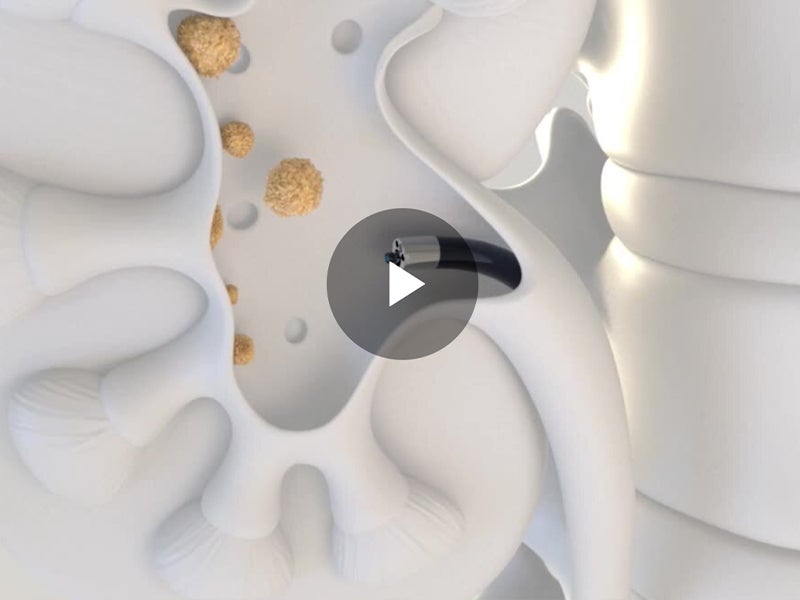
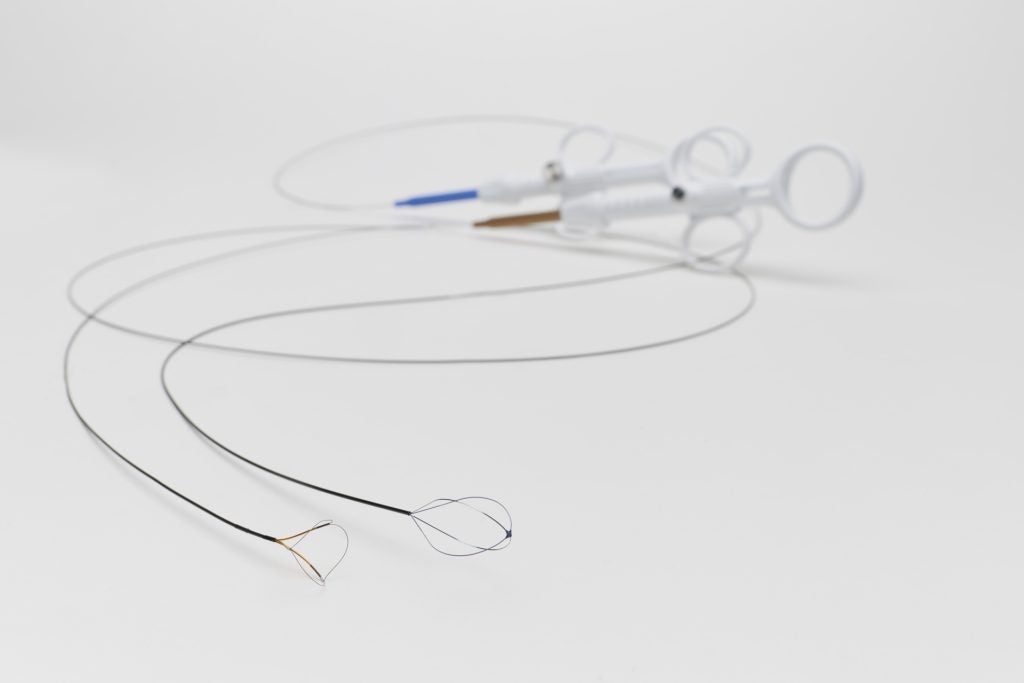
Alleima is a world-leading manufacturer of stainless steel and advanced alloys for the most demanding industries. Its medical unit is a trusted partner of leading OEMs and medical device companies to design and develop wire-based solutions for state-of-the-art medical devices. With its broad range of core and customized capabilities including fine wire drawing, nitinol braiding and shape-setting, and coating to name a few, Alleima offers tailor-made services to its client’s specific needs. Current applications span a variety of sectors, including cardiac rhythm management, cochlear, electromyography, oncology, and urology as well as orthopaedics and ophthalmology. To learn more about Alleima’s expertise in wire-based and nitinol solutions for medical devices, visit the company’s content library.

With its fully integrated value chain, Alleima provides industry-leading metallurgy technology, quality, sustainability, and circularity to support leading medical OEMs in their efforts to produce the most advanced medical components. Today Alleima offers over 200 medical alloys as standard and more on request. Alleima expanded its offering in 2023 to include processing nitinol, the shape memory alloy, that is required in numerous state-of-the-art medical devices such as robotic surgery and advanced endoscopic applications.
Industry-leading sustainability and circularity
Sustainability is present in all aspects of Alleima’s operations and is an integral part of the company’s commercial strategy. The main contribution to sustainability is through our product offering, enabling the transition to renewable energy sources, electrification of industries, innovation in the medical sector, and much more. Alleima also actively seek to minimize the impact on the environment via our operations.
Alleima reconfirmed with Ecovadis gold medal for sustainable performance in September 2025. EcoVadis is a leading provider of corporate sustainability ratings. This year, Alleima has also started to report its scope 3 emissions. This prestigious recognition of sustainable business practices places Alleima in the top 5% among the more than 150,000 companies assessed worldwide.
EcoVadis is one of the world’s most trusted providers of business sustainability ratings, intelligence, and collaborative performance improvement tools for global supply chains. The methodology used by EcoVadis is based on international sustainability standards and supervised by a scientific community of sustainability and supply chain experts. EcoVadis looks at how companies implement sustainability across their supply chain.
Alleima is also committed to a number of international initiatives, including the UN Sustainable Development Goals (SDGs) which outline the world’s common agenda for 2030. The company is committed to all 17 goals and has identified 10 that are particularly relevant for its business and where it can make the greatest impact. Read more at UN Sustainable Development Goals — Alleima
Alleima introduces AI within its research and development with Alleima Guru
Alleima has long been exploring how generative AI can be used to make operations smarter, better, and more efficient. The products that are manufactured are often used in very tough environments, which means that there are very high demands on the company’s research and development to develop the best solutions for its customers. Today, Alleima has around 900 active recipes for various alloys and approximately 250 employees work in research and development around the world, with the majority in Sandviken. Over the years, the company has collected approximately 60,000 research reports in its archive. With Alleima Guru, it will be easier and faster to get access to previous research to save time to market for new products and even better tackle challenges that our customers face and how we at Alleima can contribute to them.
To learn more about Alleima’s expertise in wire-based and nitinol solutions for medical devices, visit the company’s content library.