Abbott obtains FDA approval for first-in-class MitraClip device

Abbott has obtained US Food and Drug Administration (FDA) approval for its first-in-class, catheter-based MitraClip therapy, designed for patients with significant mitral regurgitation (MR) who have no surgical treatment options to improve their quality of life.
With FDA approval, the MitraClip device will be immediately available in the US, providing physicians with a new treatment option for patients with significant symptomatic degenerative MR who are at prohibitive risk for mitral valve surgery.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
MR is a condition involving the mitral valve of the heart no longer functioning properly, leading to a backward flow of blood.
Boston Scientific obtains clearance for bronchial thermoplasty system in China
Boston Scientific has obtained China Food and Drug Administration approval for its Alair bronchial thermoplasty system, designed to treat severe asthma.
The Alair system is unique and effective therapy intended for patients whose severe and persistent asthma is not well-controlled with inhaled corticosteroids and long-acting beta agonist medications.
Bronchial Thermoplasty (BT) is a bronchoscopic procedure typically performed under moderate sedation on an outpatient basis.
Hospira launches next-generation portable infusion pump system in US
Hospira has announced the introduction of Sapphire infusion system, a next-generation portable pump, following regulatory clearance from the US Food and Drug Administration (FDA).
It features a full-colour touchscreen and software designed to enable intuitive programming and operation with minimal steps.
The smart pump capabilities of the Sapphire system help healthcare organisations support a high-level of medication delivery safety.
Cardiovascular Systems obtains FDA approval for first new atherectomy device
US-based Cardiovascular Systems has received pre-market approval (PMA) from the US Food and Drug Administration (FDA) to market its Diamondback 360 coronary orbital atherectomy system (OAS), designed to treat severely calcified coronary arteries.
The system is electronically driven by a 1.25mm diamond-coated crown that safely eliminates calcium deposits from coronary blood vessels.
It ultimately helps enable successful stent deployment, which facilitates more favourable patient outcomes.
Novo Nordisk’s new prefilled insulin pen receives Health Canada approval

Novo Nordisk Canada has obtained Health Canada approval for its Levemir FlexTouch, a disposable prefilled insulin pen containing Levemir (insulin detemir), designed for treating patients with type 1 and type 2 diabetes.
FlexTouch is the only prefilled insulin pen that features an easy touch button and a spring-loaded mechanism.
The spring-loaded mechanism in the FlexTouch ensures that no matter how high the dose required, the push button does not extend and only a minimum of pressure is needed to deliver the dose.
St Jude Medical obtains CE Mark approval for leadless pacemaker
US-based medical technology developer St Jude Medical has acquired Nanostim and its miniature leadless pacemaker, following European approval of the device.
The Nanostim pacemaker is implanted using a steerable catheter through the femoral vein in a minimally invasive procedure, eliminating the need to surgically create a room for the pacemaker and insulated wires.
It is a modification over existing models that the company stresses is a major safety improvement.
Samsung launches first tablet-based ultrasound device
Samsung Electronics America has launched UGEO PT60A, its first tablet-based ultrasound system for the point-of-care (POC) market, enabling doctors to perform medical diagnoses at or near the site of patient care.
The UGEO PT60A is a compact device that combines highly efficient ultrasound functions for the purpose of POC.
UGEO PT60A features the latest technologies, such as Needle Mate for precise identification of the needle’s location, SDMR providing clearer imaging with an innovative noise reduction filter and SCI, delivering remarkable spatial and contrast resolution.
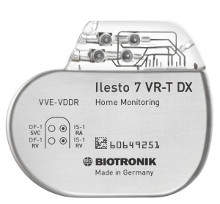
Biotronik launches new Ilesto implantable cardiac defibrillator
Biotronik, a German medical device company, has launched its next-generation technology platform, Ilesto DX, following FDA approval for its Ilesto family of implantable cardioverter-defibrillator / cardiac resynchronisation therapy defibrillators (ICD/CRT-D).
The new electromechanical platform is 15% smaller in size and requires 29% fewer components, with no compromise in longevity or clinical features.
It is reportedly the first defibrillator system to use only one specialised defibrillator lead to provide full atrial diagnostic information.






