Jarvik Heart obtains regulatory clearance for Jarvik 2000 Heart in Japan
Jarvik Heart obtained regulatory approval for its Jarvik 2000 Heart, a left ventricular assist device (LVAD) implanted inside the heart, from the Japanese Pharmaceuticals and Medical Devices Agency (PMDA).
On November 22, Shonin approval was granted based on Japanese and US clinical trial data of the device, approving it as a bridge to transplant. Century Medical will be the exclusive distributor of Jarvik 2000 in Japan.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
At only 30cc volume and 90g (about the size of a C-cell battery), the Jarvik 2000 can support the hearts of patients only 4ft 6in tall and weighing less than 85lb.
Grifols to buy Novartis’s diagnostics business for $1.7bn
Spanish company Grifols announced a definitive agreement to acquire Novartis’s blood transfusion diagnostics unit for $1.675bn.
The transaction will give critical mass and a significant US presence to Grifols’s previously small diagnostics business, which in future is expected to account for more than 20% of the company’s sales, compared with the current 4%.
The deal will be structured through Grifols’s diagnostic division and a newly created 100% Grifols-owned subsidiary. It will also see an increase in Grifols’ workforce by approximately 550 employees as former Novartis employees are likely to be retained.
Abbott obtains FDA approval for first-in-class MitraClip device

Abbott obtained US Food and Drug Administration (FDA) approval for its first-in-class, catheter-based MitraClip therapy, designed for patients with significant mitral regurgitation (MR) who have no surgical treatment options to improve their quality of life.
With FDA approval, the MitraClip device will be immediately available in the US, providing physicians with a new treatment option for patients with significant symptomatic degenerative MR who are at prohibitive risk for mitral valve surgery.
MR is a condition involving the mitral valve of the heart no longer functioning properly, leading to a backward flow of blood.
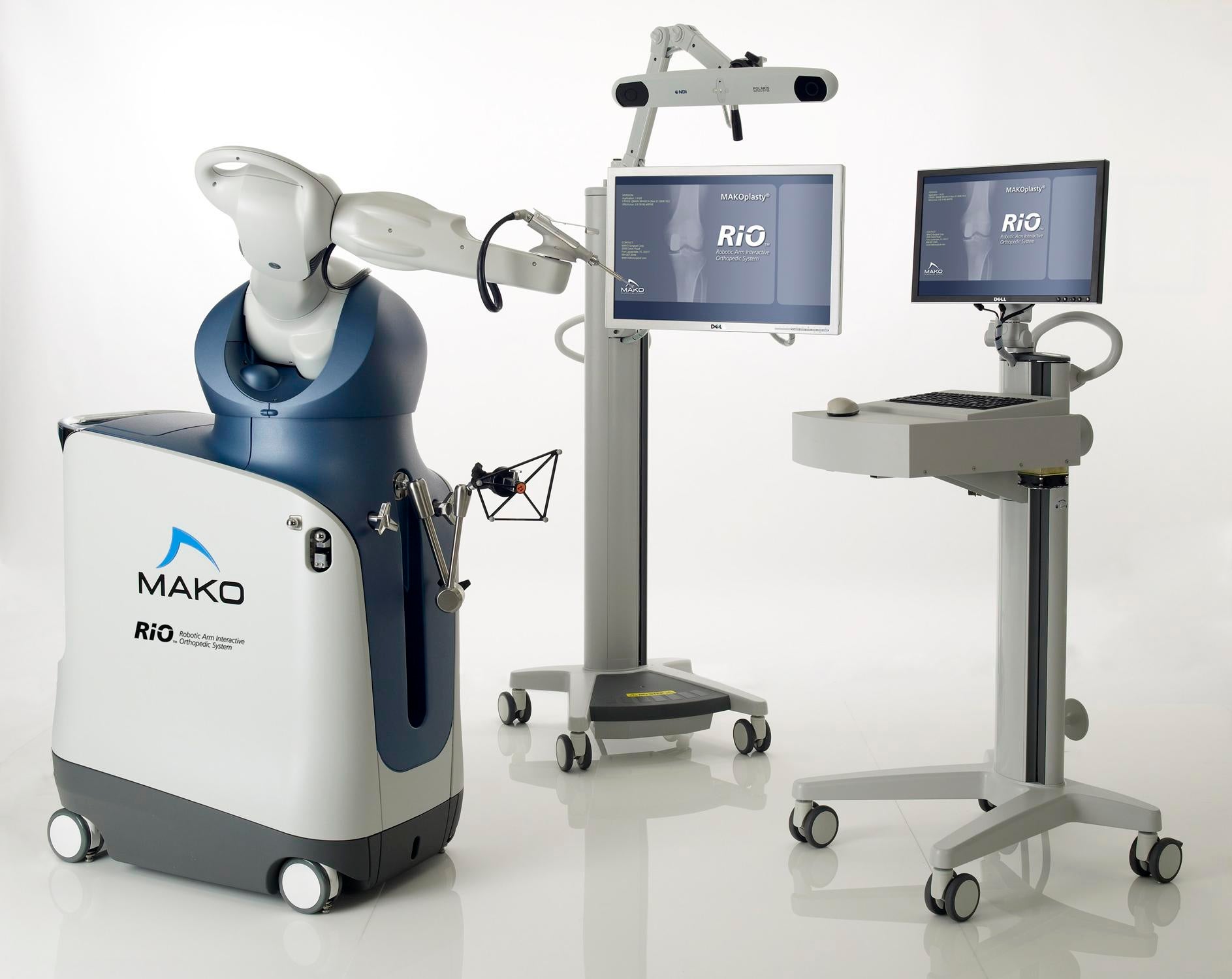
Stryker to acquire Mako Surgical for $1.65bn
US medical equipment manufacturer Stryker signed a definitive agreement to acquire Mako Surgical, developer of robotic solution and implants for minimally invasive orthopaedic procedures, for $30 per share with an aggregate purchase price of approximately $1.65bn.
Mako Surgical markets joint-specific applications for the knee and hip, proprietary Restoris implants for orthopaedic procedures called MAKOplasty, and the Rio robotic arm interactive orthopaedic system.
The Rio is a surgeon-interactive tactile surgical platform that incorporates a robotic arm and patient-specific visualisation technology, which enables precise, consistently reproducible bone resection for the accurate insertion and alignment of Mako’s Restoris implants.
Given Imaging’s PillCam SB 3 video capsule receives 510(k) clearance
Israel-based medical devices developer Given Imaging received clearance from the US regulatory agency to market its next-generation video capsule PillCam SB 3, which can detect small bowel abnormalities related to Crohn’s disease and gastrointestinal bleeding.
Weighing less than 4g, the 11mm by 26mm video capsule features an imaging device and a light source that can transmit anywhere between two and six images in one second.
Due to software enhancements, the PillCam SB 3 offers a 40% more efficient video compilation than the earlier SB 2 version. The SB 3 will be available in the US towards the end of the year.
Scientists develop intelligent surgical knife to detect cancerous tissue
Researchers at Imperial College London in the UK created an intelligent knife, which can help surgeons identify cancerous tissue.
The iKnife is based on an electrosurgery technology invented in the 1920s and widely used in the medical field today.
With the help of an electrical current, an electrosurgical knife can quickly heat the tissue and then cut through it, while simultaneously reducing the loss of blood.
UK’s MHRA to regulate electronic cigarettes as medicines
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) is to regulate all products containing nicotine, including electronic cigarettes, as medicines to ensure product safety and reduce the negative effects of smoking.
The regulation follows concerns that an increase in the popularity of e-cigarettes had led to unregulated and potentially unsafe products being sold.
In England, smoking is the largest single cause of avoidable death every year, killing 80,000 people, and it is hoped that making safe and effective products available for smokers will reduce this risk.
Valeant to acquire Bausch + Lomb for $8.7bn
Canada’s leading pharmaceutical company Valeant Pharmaceuticals entered into a definitive agreement to acquire Bausch + Lomb Holdings, a US-based supplier of eye health products, for $8.7bn.
Bausch + Lomb provides vision care products such as contact lenses and solutions; surgical devices such as intraocular lenses and surgical equipment and pharmaceutical drugs such as prescription brands, generics and over-the-counter (OTC) drugs.
The acquisition is a part of Valeant’s strategy to take advantage of the growing demand for eye health products, which is driven by an aging patient population, an increased rate of diabetes and demand from emerging markets.
St Jude wins CE mark for deep brain stimulation systems

The European Union granted CE mark certification to St Jude Medical’s deep brain stimulation (DBS) systems, designed for the treatment of primary and secondary dystonia, a neurological movement disorder.
The devices, Brio, Libra and LibraXP, are placed using two thin leads with small electrodes that are connected to a power source placed just under the skin near the pectoral muscle in the chest.
The DBS therapy delivers a constant series of painless electrical pulses to the specific target regions including the subthalamic nucleus (STN) or the globus pallidus interna (GPi) of the brain, which are involved with controlling movement.
Medtronic wins CE mark for Engager transcatheter aortic valve implant
The European Union granted CE mark approval to Medtronic’s new transcatheter aortic valve implantation (TAVI) system with transapical delivery catheter, for the treatment of patients with severe aortic stenosis who are at high or extreme risk of surgical aortic valve replacement (SAVR).
Using a minimally-invasive delivery system via a catheter, the Engager TAVI system is inserted in the apex (the lower, pointed end) of the heart to promote annular sealing and minimise paravalvular leak (PVL).
Comprising bovine tissue leaflets and a self-expanding nitinol frame, the valve’s control arms simplify implantation, and the supra-annular valve positioning facilitates leaflet coaptation (connections) in non-circular anatomy for optimal hemodynamic performance, according to the company.
Biotronik wins CE mark for Ilesto 7 CD/CRT-D series

The European Union granted CE mark certification to Biotronik’s Ilesto 7 implantable cardioverter-defibrillator / cardiac resynchronisation therapy defibrillator (ICD/CRT-D) series.
The Ilesto 7 series, which the company claims include the world’s smallest ICDs, feature the company’s ProMRI technology, which enables access to potentially life-saving MR scans.
Lucerne Cantonal Hospital physician Dr Paul Erne said patients often require MR scanning at some point in the future, as they get older and have comorbidities.
Direct Flow Medical wins CE mark for transcatheter aortic heart valve system
The European Union granted CE mark certification to Direct Flow Medical’s transcatheter aortic heart valve system, featuring a double-ring design to create a tight and durable seal around the annulus.
Using pressurised saline and contrast solution, the Direct Flow Medical transcatheter aortic heart valve’s polymer-based metal-free frame is expanded for placement, assessment and repositioning, according to the company.
The system also eliminates aortic regurgitation and reduces procedural risk by eliminating the need for rapid pacing during deployment and post-dilitation.
Related content
The evolution of e-labelling in Europe
The adoption of e-labelling, the inclusion of digital instructions with medical devices, has been very slow in Europe. Medical Device Developments spoke to Dario Pirovano, consultant for industry body Eucomed, about the benefits of e-labelling and whether regulatory reform will increasingly promote its use in Europe.