Case Study: Epionics Corporate Branding

Erdmann Design offered a comprehensive corporate branding service to start-up company Epionics for the introduction of its new Epionics Spine Monitoring System, including product design, brand design, screen design and corporate communication.
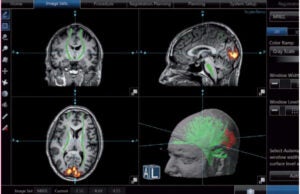
The Epionics System is a 24-hour monitoring device for the spine. It is designed to help reduce the number of chronic back problems and high complication rates.
This new gold standard for diagnosis and treatment validation of lower back pain was developed through human-centred design methods by Erdmann Design. After a practice-related analysis of the product idea, the examination of company values revealed the strategy for corporate branding and led to the company’s development.
Cooperation with specific partners led to the creation of risk and monitoring validation using design thinking models with the new medical standard in pain therapy. The advantages of the Epionics Spine Monitoring System can be implemented immediately by doctors and healthcare specialists in 24-hour movement measurement.