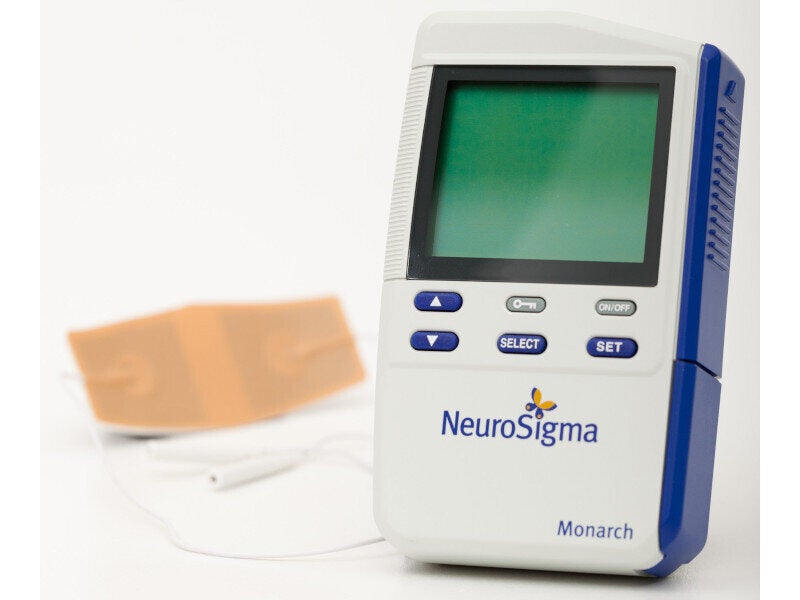
NeuroSigma’s Monarch external trigeminal nerve stimulation (eTNS) system is a non-pharmaceutical monotherapy for the treatment of attention deficit hyperactivity disorder (ADHD) in paediatric patients aged seven years to 12 years.
It is a prescription-only device and the company’s first non-drug treatment for ADHD patients. It is designed for at-home use under the supervision of physicians.
Regulatory approvals for Monarch eTNS system
The Monarch eTNS system was first approved in Canada in May 2013 as a Class II medical device for drug-resistant epilepsy (DRE), major depressive disorder, and treatment-resistant depression.
The Australian Therapeutic Goods Administration approved the device in April 2014 as a supplementary treatment for DRE in patients aged nine years and older. The device received humanitarian use device designation from the US Food and Drug Administration (FDA) for the treatment of Lennox-Gastaut Syndrome symptoms in January 2015.
In addition, the Monarch eTNS system received the European CE mark in November 2015 and FDA clearance in April 2019 for the treatment of paediatric ADHD. The FDA approved the device via the de novo pathway under the classification name Transcutaneous Nerve Stimulator for ADHD.
NeuroSigma teamed up with Quick Care Pharmacy, a speciality pharmacy in California, US, for the sales and distribution of the Monarch eTNS system in the US, in October 2019.
In January 2022, NeuroSigma provided exclusive rights to Ignis Therapeutics, a life sciences company based in China, for the development and commercialisation of the Monarch eTNS system in China, as well as in Hong Kong and Macau.
In February 2022, the Monarch eTNS system was granted FDA breakthrough device designation for use as an adjunct for the reduction of seizures in patients aged 18 years and older diagnosed with epilepsy, characterised by partial-onset seizures with or without secondary generalisation that are refractory to two or more anti-epileptic medications. The device was recognised as a novel device for DRE and a safer and more effective alternative to drugs or surgery for a growing number of severe conditions.
The Monarch eTNS was launched commercially in October 2022.
Monarch eTNS system features
Monarch eTNS is a non-invasive, wearable therapy that eliminates many of the risks associated with drugs, implants, and brain surgery.
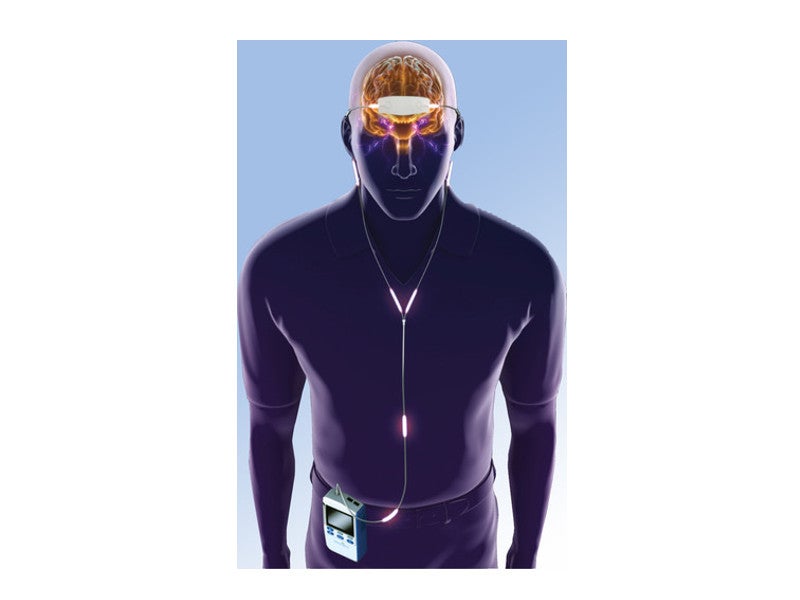
It is a mild nerve-stimulating device, comprising a portable pulse generator connected to a single-use electric patch, which is placed on the patient’s forehead.
Low-level electric signals are transmitted to the electrode patches, which stimulate trigeminal nerve fibres and send therapeutic signals to the areas of the brain known to be involved with ADHD.
Monarch eTNS mechanism of action
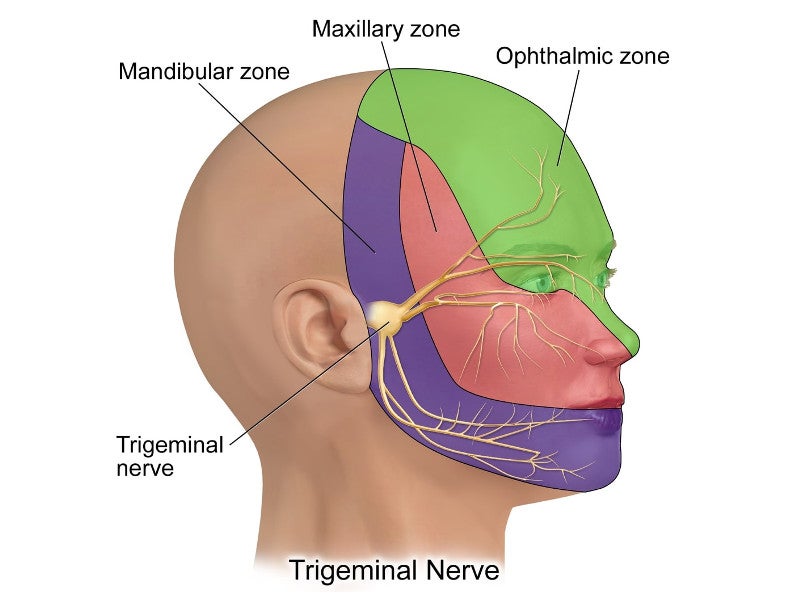
The trigeminal nerve is the fifth and largest cranial nerve. It is directly connected to the brain and controls facial sensation. The nerve either directly or indirectly enters the regions of the brain associated with ADHD and other disorders.
eTNS provides a mild electrical pulse to various branches of the trigeminal nerve and tends to increase the activity in the regions of the brain associated with regulating attention and behaviour.
Clinical trials on Monarch eTNS system
The FDA’s approval of the device was based on a pivotal, double-blind, randomised controlled trial conducted at the University of California, Los Angeles, US. A total of 62 children with moderate-to-severe ADHD were enrolled in the trial.
The device was compared with placebo and used on patients for four weeks. The primary endpoint of the trial was improvement in the ADHD-Rating Scale IV score, which is used to measure the severity of ADHD symptoms. A higher score on the scale indicates worsening symptoms.
At week four, children treated with the device demonstrated a decrease in ADHD-RS-IV score from 34.1 points to 23.4 points compared to a decrease from 33.7 points to 27.5 points in the placebo group. The safety profile of the device was also favourable.
Significant improvement in the clinical global impression improvement scale was also observed in patients treated with the device compared to the placebo group.
Common side effects reported during the study were trouble sleeping, improved appetite, headache, and teeth clenching.
Market commentary on NeuroSigma
Based in the US, NeuroSigma is a life sciences company involved in the development and marketing of bioelectronic technologies such as eTNS.
The company’s eTNS neuromodulation therapies are developed on the intellectual property exclusively licensed from the University of California. The therapies cover a broad range of disorders such as ADHD, epilepsy, and post-traumatic stress disorder.
Meanwhile, NeuroSigma’s eTNS platform and Monarch system’s pipeline indications include a broad range of neurological and neuropsychiatric disorders such as ADHD, drug-resistant epilepsy, and depression.